Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
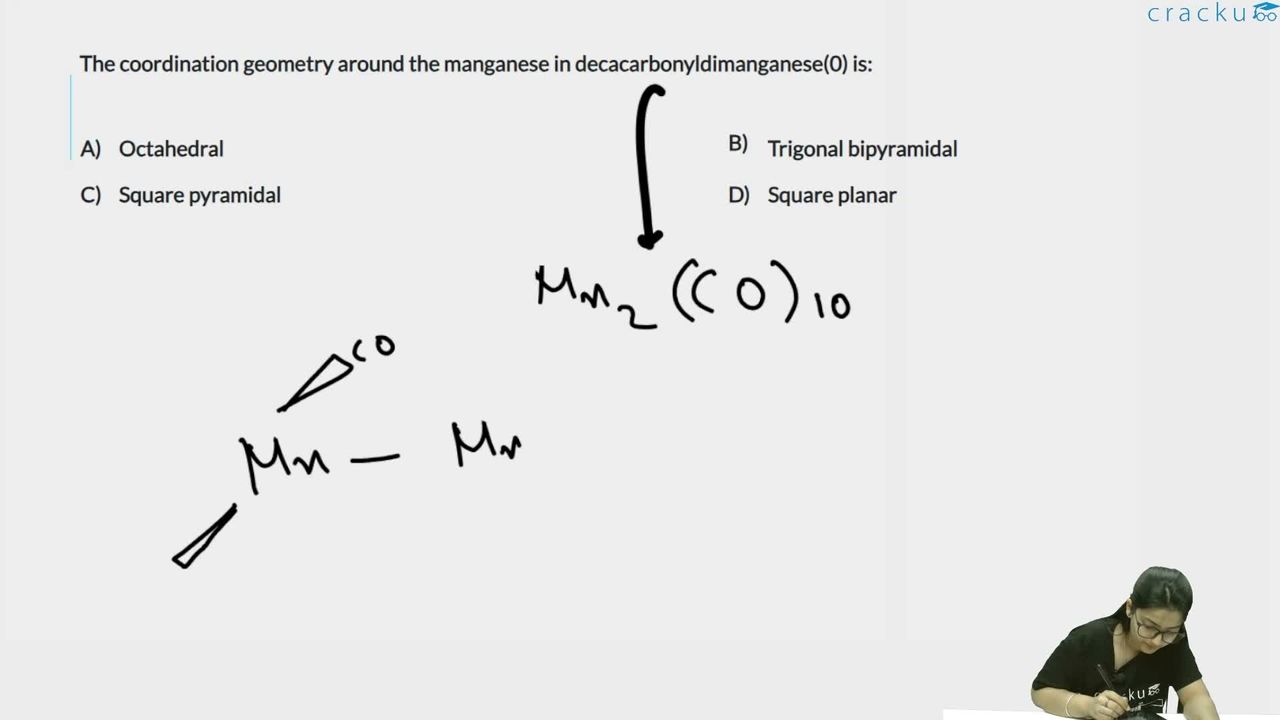
Decacarbonyldimanganese(0) is Mn$$_2$$(CO)$$_{10}$$.
Each Mn is bonded to 5 CO ligands and one Mn-Mn bond, giving 6 groups around each Mn.
This corresponds to octahedral geometry.
The answer is Option (1): Octahedral.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
