NTA JEE Main 9th January 2019 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 31
For emission line of atomic hydrogen from $$n_i = 8$$ to $$n_f = n$$, the plot of wave number $$\bar{\nu}$$ against $$\frac{1}{n^2}$$ will be: (The Rydberg constant, $$R_H$$ is in wave number unit)
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 32
In general, the properties that decrease and increase down a group in the periodic table, respectively, are:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 33
Aluminium is usually found in +3 oxidation state. In contrast, thallium exists in +1 and +3 oxidation states. This is due to:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 34
According to molecular orbital theory, which of the following is true with respect to $$Li_2^+$$ and $$Li_2^-$$?
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 35
0.5 moles of gas A and x moles of gas B exert a pressure of 200 Pa in a container of volume 10 m$$^3$$ at 1000 K. Given, R is the gas constant in JK$$^{-1}$$mol$$^{-1}$$, x is:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 36
Consider the reversible isothermal expansion of an ideal gas in a closed system at two different temperatures $$T_1$$ and $$T_2$$ ($$T_1 < T_2$$). The correct graphical depiction of the dependence of work done $$w$$ vs the final volume $$V$$ is:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 37
20 ml of 0.1 M $$H_2SO_4$$ solution is added to 30 mL of 0.2 M $$NH_4OH$$ solution. The pH of the resultant mixture is: ($$pK_b$$ of $$NH_4OH = 4.7$$)
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 38
The isotopes of hydrogen are:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 39
The alkaline earth metal nitrate that does not crystallise with molecules is:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 40
Correct statements among regarding silicones are:
(a) They are polymers with hydrophobic character.
(b) They are biocompatible.
(c) In general, they have high thermal stability and low dielectric strength.
(d) Usually, they are resistant to oxidation and used as greases.
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 41
Arrange the following amines in the decreasing order of basicity.
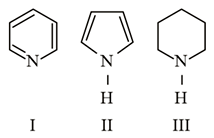
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 42
Which amongst the following is the strongest acid?
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 43
The compounds A and B in the following reaction are, respectively:
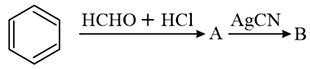
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 44
A water sample has ppm level concentration of the following metals: Fe = 0.2; Mn = 5.0; Cu = 3.0; Zn = 5.0. The metal that makes the water sample unsuitable for drinking is:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 45
The one that is extensively used as a piezoelectric material is:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 46
A solution of sodium sulphate contains 92 g of Na$$^+$$ ions per kilogram of water. The molality of Na$$^+$$ ions in that solution in mol kg$$^{-1}$$ is:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 47
Which one of the following statements regarding Henry's law is not correct?
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 48
The anodic half-cell of lead-acid battery is recharged using electricity of 0.05 Faraday. The amount of $$PbSO_4$$ electrolyzed in g during the process is: (Molar mass of $$PbSO_4 = 303$$ g mol$$^{-1}$$)
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 49
The following results were obtained during kinetic studies of the reaction.
$$2A + B \rightarrow$$ product
Experiment I: A = 0.10 mol L$$^{-1}$$, B = 0.20 mol L$$^{-1}$$, Rate = $$6.93 \times 10^{-3}$$ mol L$$^{-1}$$ min$$^{-1}$$
Experiment II: A = 0.10 mol L$$^{-1}$$, B = 0.25 mol L$$^{-1}$$, Rate = $$6.93 \times 10^{-3}$$ mol L$$^{-1}$$ min$$^{-1}$$
Experiment III: A = 0.20 mol L$$^{-1}$$, B = 0.30 mol L$$^{-1}$$, Rate = $$1.386 \times 10^{-2}$$ mol L$$^{-1}$$ min$$^{-1}$$
The time (in minutes) required to consume half of A is:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 50
Adsorption of a gas follows Freundlich adsorption isotherm. In the given plot, $$x$$ is the mass of the gas adsorbed on mass $$m$$ of the adsorbent at pressure P. $$\frac{x}{m}$$ is proportional to:
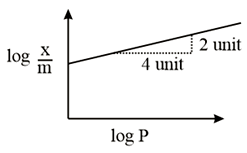
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 51
The ore that contains both iron and copper is:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 52
The major product of the following reaction is:
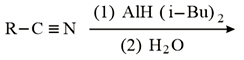
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 53
Two complexes $$[CrH_2O)_6]Cl_3$$ (A) and $$[Cr(NH_3)_6]Cl_3$$ (B) are violet and yellow coloured, respectively. The incorrect statement regarding them is:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 54
In the reaction given below what will be the major product at the end:
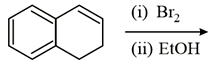
Options:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 55
Major product of the given reaction is:
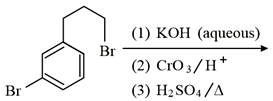
Options:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 56
The highest value of the calculated spin only magnetic moment (in BM) among all the transition metal complexes is:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 57
Acid strength given below the correct decreasing order will be:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 58
Major product of the given below reaction:
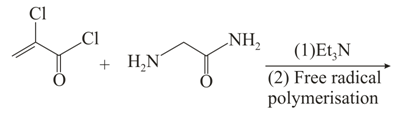
Options:
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 59
The correct match between column-I and column-II is:
Column-I (drug) Column-II (test)
(A) Chloroxylenol (P) Carbylamine test
(B) Norethindrone (Q) Sodium hydrogen carbonate test
(C) Sulphapyridine (R) Ferric chloride test
(D) Penicillin (S) Baeyer's test
NTA JEE Main 9th January 2019 Shift 1 - Chemistry - Question 60
The increasing order of pKa of the following amino acids in aqueous solution is: Glycine, Aspartate, Lysine, Arginine.
.webp)
.webp)
.webp)
.webp)




