Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Adsorption of a gas follows Freundlich adsorption isotherm. In the given plot, $$x$$ is the mass of the gas adsorbed on mass $$m$$ of the adsorbent at pressure P. $$\frac{x}{m}$$ is proportional to:
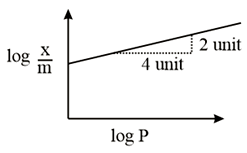
The line is y = mx + c. Here the slope is 2/4 = 1/2. Hence on taking anti log on b/s we see that x/m is directly proportional to p^1/2
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
