NTA JEE Mains 06th April 2024 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 31
Molality ($$m$$) of $$3M$$ aqueous solution of NaCl is : (Given : Density of solution $$= 1.25 \text{ g mL}^{-1}$$, Molar mass in $$\text{gmol}^{-1}$$: Na $$- 23$$, Cl $$- 35.5$$)
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 32
The ratio $$\frac{K_P}{K_C}$$ for the reaction : $$CO_{(g)} + \frac{1}{2} O_{2(g)} \rightleftharpoons CO_{2(g)}$$ is :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 33
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 34
The number of ions from the following that are expected to behave as oxidising agent is : $$Sn^{4+}, Sn^{2+}, Pb^{2+}, Tl^{3+}, Pb^{4+}, Tl^{+}$$
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 35
Evaluate the following statements related to group 14 elements for their correctness. (A) Covalent radius decreases down the group from C to Pb in a regular manner. (B) Electronegativity decreases from C to Pb down the group gradually. (C) Maximum covalance of C is 4 whereas other elements can expand their covalance due to presence of d orbitals. (D) Heavier elements do not form $$p\pi - p\pi$$ bonds. (E) Carbon can exhibit negative oxidation states. Choose the correct answer from the options given below :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 36
The correct statement among the following, for a "chromatography" purification method is :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 37
The incorrect statement regarding the geometrical isomers of 2-butene is :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 38
How can an electrochemical cell be converted into an electrolytic cell?
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 39
Arrange the following elements in the increasing order of number of unpaired electrons in it. (A) Sc (B) Cr (C) V (D) Ti (E) Mn. Choose the correct answer from the options given below :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 40
The correct IUPAC name of $$[PtBr_2(PMe_3)_2]$$ is :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 41
Given below are two statements : Statement I : $$PF_5$$ and $$BrF_5$$ both exhibit $$sp^3d$$ hybridisation. Statement II : Both $$SF_6$$ and $$[Co(NH_3)_6]^{3+}$$ exhibit $$sp^3d^2$$ hybridisation. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 42
Match List - I with List - II.

Choose the correct answer from the options given below :
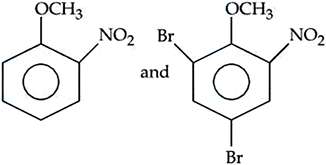
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 43
The correct arrangement for decreasing order of electrophilic substitution for above compounds is :
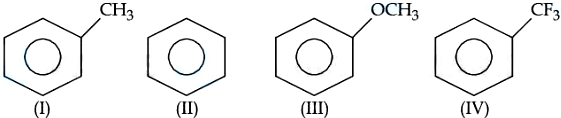
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 44
Consider the given reaction, identify the major product "P". $$CH_3 - COOH \xrightarrow{(i) LiAlH_4 \; (ii) PCC \; (iii) HCN/OH^{-} \; (iv) H_2O/OH^{-}, \Delta}$$ "P"
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 45
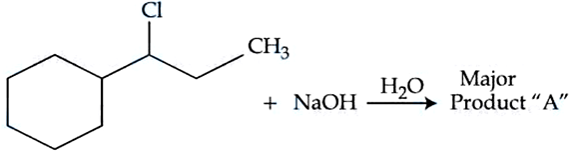
Consider the above chemical reaction. Product "A " is :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 46
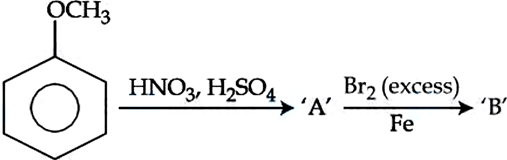
The major products formed A and B respectively are:
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 47
Identify the product (A) in the following reaction.
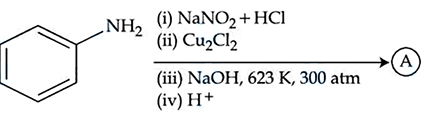
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 48
During the detection of acidic radical present in a salt, a student gets a pale yellow precipitate soluble with difficulty in $$NH_4OH$$ solution when sodium carbonate extract was first acidified with dil. $$HNO_3$$ and then $$AgNO_3$$ solution was added. This indicates presence of :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 49
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 50
The incorrect statements regarding enzymes are : (A) Enzymes are biocatalysts. (B) Enzymes are non-specific and can catalyse different kinds of reactions. (C) Most Enzymes are globular proteins. (D) Enzyme - oxidase catalyses the hydrolysis of maltose into glucose. Choose the correct answer from the option given below :
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 51
Consider the following reactions $$NiS + HNO_3 + HCl \rightarrow A + NO + S + H_2O$$. $$A + NH_4OH + H_3C - C(=N-OH) - C(=N-OH) - H_3C \rightarrow B + NH_4Cl + H_2O$$. The number of protons that do not involve in hydrogen bonding in the product $$B$$ is ___________.
789
456
123
0.-
Clear All
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 52
For hydrogen atom, energy of an electron in first excited state is $$-3.4 \text{ eV}$$, K.E. of the same electron of hydrogen atom is $$x \text{ eV}$$. Value of $$x$$ is ___________ $$\times 10^{-1} \text{ eV}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 53
An amine (X) is prepared by ammonolysis of benzyl chloride. On adding p-toluenesulphonyl chloride to it the solution remains clear. Molar mass of the amine (X) formed is ___________ $$\text{gmol}^{-1}$$. (Given molar mass in $$\text{gmol}^{-1}$$: C : 12, H : 1, O : 16, N : 14)
789
456
123
0.-
Clear All
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 54
For the reaction at $$298 \text{ K}$$, $$2A + B \rightarrow C$$. $$\Delta H = 400 \text{ kJ mol}^{-1}$$ and $$\Delta S = 0.2 \text{ kJ mol}^{-1} \text{K}^{-1}$$. The reaction will become spontaneous above ___________ K.
789
456
123
0.-
Clear All
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 55
Consider the two different first order reactions given below $$A + B \rightarrow C$$ (Reaction 1) $$P \rightarrow Q$$ (Reaction 2). The ratio of the half life of Reaction 1 : Reaction 2 is $$5 : 2$$. If $$t_1$$ and $$t_2$$ represent the time taken to complete $$\frac{2}{3}^{rd}$$ and $$\frac{4}{5}^{th}$$ of Reaction 1 and Reaction 2, respectively, then the value of the ratio $$t_1 : t_2$$ is ___________ $$\times 10^{-1}$$ (nearest integer). [Given : $$\log_{10}(3) = 0.477$$ and $$\log_{10}(5) = 0.699$$]
789
456
123
0.-
Clear All
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 56
Among $$VO_2^{+}$$, $$MnO_4^{-}$$ and $$Cr_2O_7^{2-}$$, the spin-only magnetic moment value of the species with least oxidising ability is ___________ BM (Nearest integer). (Given atomic number $$V = 23, Mn = 25, Cr = 24$$)
789
456
123
0.-
Clear All
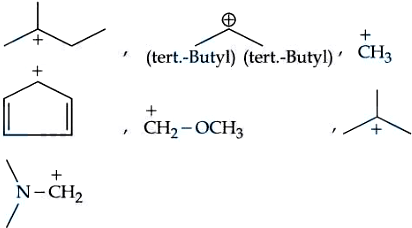
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 57
Number of carbocations from the following that are not stabilized by hyperconjugation is __________
_
789
456
123
0.-
Clear All
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 58
When '$$x$$' $$\times 10^{-2}$$ mL methanol (molar mass $$= 32 \text{ g}$$; density $$= 0.792 \text{ g/cm}^3$$) is added to $$100 \text{ mL}$$ water (density $$= 1 \text{ g/cm}^3$$), the following diagram is obtained. $$x =$$ ___________ (nearest integer).
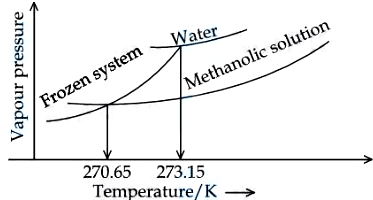
[Given : Molal freezing point depression constant of water at $$273.15 \text{ K}$$ is $$1.86 \text{ K kg mol}^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 59
Total number of species from the following with central atom utilising $$sp^2$$ hybrid orbitals for bonding is ___________. $$NH_3$$, $$SO_2$$, $$SiO_2$$, $$BeCl_2$$, $$C_2H_2$$, $$C_2H_4$$, $$BCl_3$$, $$HCHO$$, $$C_6H_6$$, $$BF_3$$, $$C_2H_4Cl_2$$
789
456
123
0.-
Clear All
NTA JEE Mains 06th April 2024 Shift 2 - Chemistry - Question 60
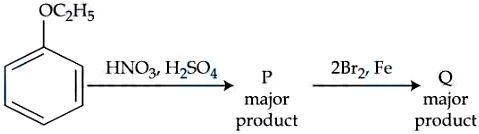
The ratio of number of oxygen atoms to bromine atoms in the product Q is ___________ $$\times 10^{-1}$$.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




