Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
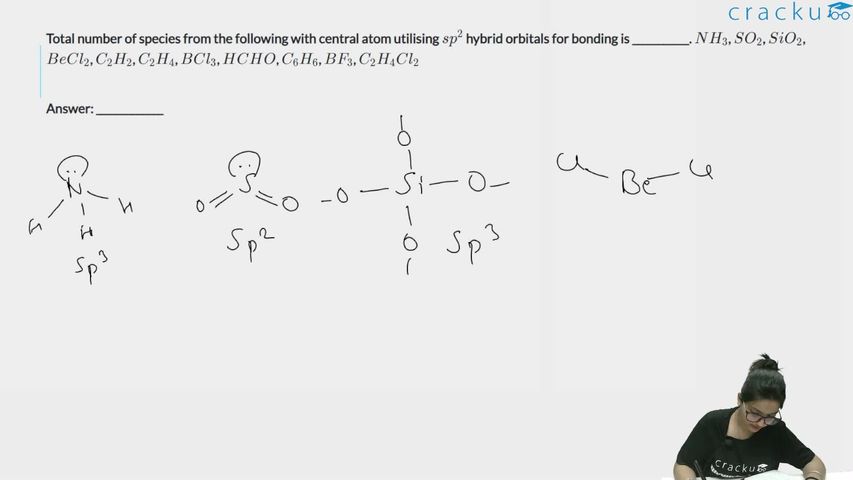
Total number of species from the following with central atom utilising $$sp^2$$ hybrid orbitals for bonding is ___________. $$NH_3$$, $$SO_2$$, $$SiO_2$$, $$BeCl_2$$, $$C_2H_2$$, $$C_2H_4$$, $$BCl_3$$, $$HCHO$$, $$C_6H_6$$, $$BF_3$$, $$C_2H_4Cl_2$$
Correct Answer: 6
We need to count the species with the central atom using sp$$^2$$ hybrid orbitals from the given list.
sp$$^2$$ hybridisation occurs when the central atom has 3 electron domains (regions of electron density), giving a trigonal planar arrangement (or bent if lone pairs are present).
Analyze each species:
(i) $$NH_3$$: N has 3 bond pairs + 1 lone pair = 4 electron domains → sp$$^3$$. ✗
(ii) $$SO_2$$: S has 2 bond pairs + 1 lone pair = 3 electron domains → sp$$^2$$. ✓
(iii) $$SiO_2$$: Each Si is bonded to 4 O atoms in a tetrahedral arrangement → sp$$^3$$. ✗
(iv) $$BeCl_2$$: Be has 2 bond pairs + 0 lone pairs = 2 electron domains → sp. ✗
(v) $$C_2H_4$$: Each C has 3 electron domains (2 C-H + 1 C=C) → sp$$^2$$. ✓
(vi) $$C_2H_2$$: Each C has 2 electron domains (1 C-H + 1 C≡C) → sp. ✗
(vii) $$BCl_3$$: B has 3 bond pairs + 0 lone pairs = 3 electron domains → sp$$^2$$. ✓
(viii) $$HCHO$$: C has 3 electron domains (2 C-H + 1 C=O) → sp$$^2$$. ✓
(ix) $$C_6H_6$$: Each C has 3 electron domains → sp$$^2$$. ✓
(x) $$BF_3$$: B has 3 bond pairs + 0 lone pairs → sp$$^2$$. ✓
(xi) $$C_2H_4Cl_2$$: Each C has 4 electron domains → sp$$^3$$. ✗
Count: SO$$_2$$, C$$_2$$H$$_4$$, BCl$$_3$$, HCHO, C$$_6$$H$$_6$$, BF$$_3$$ = 6 species.
The answer is 6.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
