Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
When '$$x$$' $$\times 10^{-2}$$ mL methanol (molar mass $$= 32 \text{ g}$$; density $$= 0.792 \text{ g/cm}^3$$) is added to $$100 \text{ mL}$$ water (density $$= 1 \text{ g/cm}^3$$), the following diagram is obtained. $$x =$$ ___________ (nearest integer).
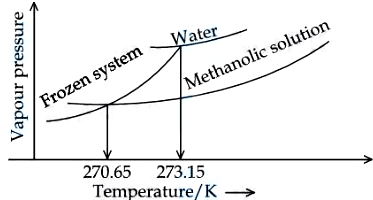
[Given : Molal freezing point depression constant of water at $$273.15 \text{ K}$$ is $$1.86 \text{ K kg mol}^{-1}$$]
Correct Answer: 543

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
