Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
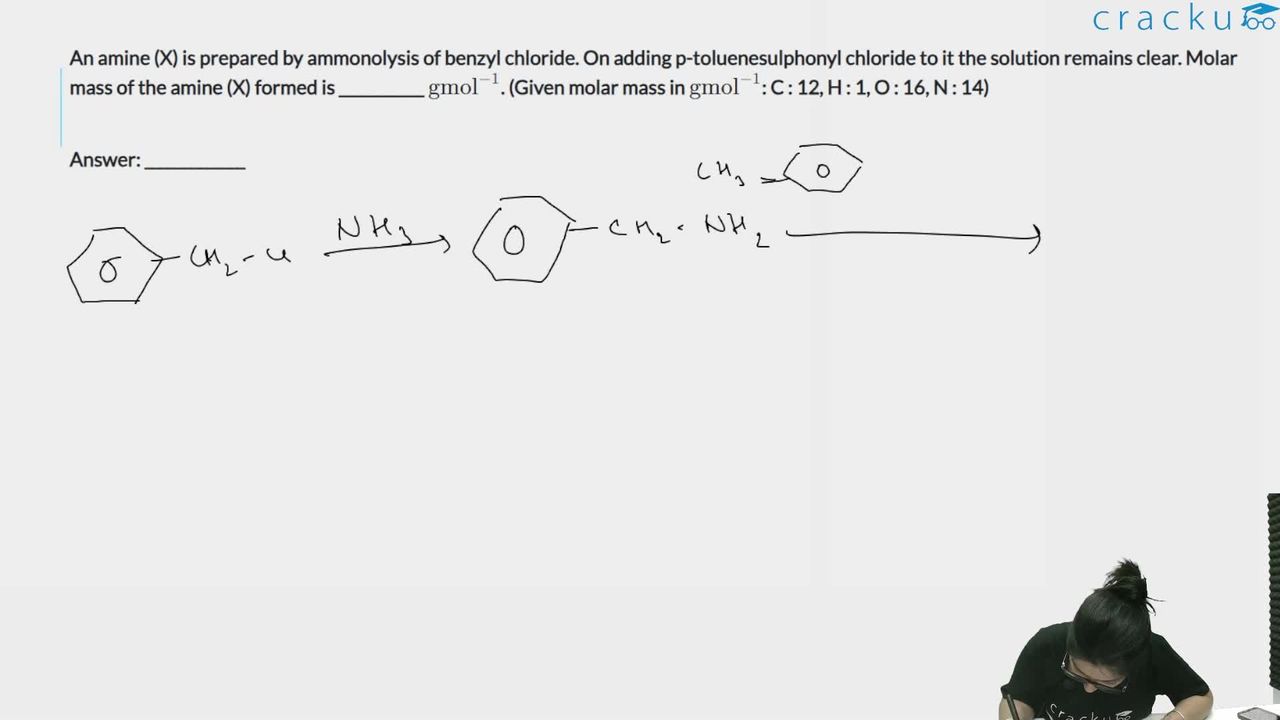
An amine (X) is prepared by ammonolysis of benzyl chloride. On adding p-toluenesulphonyl chloride to it the solution remains clear. Molar mass of the amine (X) formed is ___________ $$\text{gmol}^{-1}$$. (Given molar mass in $$\text{gmol}^{-1}$$: C : 12, H : 1, O : 16, N : 14)
Correct Answer: 287
We need to find the molar mass of amine X, prepared by ammonolysis of benzyl chloride, that gives a clear solution with the Hinsberg reagent (p-toluenesulphonyl chloride).
The Hinsberg test distinguishes between primary, secondary, and tertiary amines: (i) Primary amines react with p-toluenesulphonyl chloride to form a sulphonamide that is soluble in NaOH (acidic NH, dissolves in alkali). (ii) Secondary amines form a sulphonamide that is insoluble in NaOH (precipitate). (iii) Tertiary amines do not react — the solution remains clear.
Since the solution remains clear with p-toluenesulphonyl chloride, the amine X is a tertiary amine.
In ammonolysis of benzyl chloride (C$$_6$$H$$_5$$CH$$_2$$Cl) with excess benzyl chloride, the following sequence occurs: $$C_6H_5CH_2Cl + NH_3 \rightarrow C_6H_5CH_2NH_2$$ (primary), then $$C_6H_5CH_2Cl + C_6H_5CH_2NH_2 \rightarrow (C_6H_5CH_2)_2NH$$ (secondary), and finally $$C_6H_5CH_2Cl + (C_6H_5CH_2)_2NH \rightarrow (C_6H_5CH_2)_3N$$ (tertiary).
Therefore, the tertiary amine is tribenzylamine: $$(C_6H_5CH_2)_3N$$.
The molecular formula of tribenzylamine is $$C_{21}H_{21}N$$, and substituting into the molar mass calculation gives $$M = 21(12) + 21(1) + 14 = 252 + 21 + 14 = 287 \, \text{g/mol}$$.
Option A: 287 g mol$$^{-1}$$.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
