NTA JEE Mains 05th April 2024 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 31
The number of moles of methane required to produce $$11$$ g $$CO_2(g)$$ after complete combustion is : (Given molar mass of methane in $$g mol^{-1} : 16$$)
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 32
Given below are two statements : Statement I : The metallic radius of Na is $$1.86$$ Å and the ionic radius of $$Na^+$$ is lesser than $$1.86$$ Å. Statement II : Ions are always smaller in size than the corresponding elements. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 33
Match List I with List II :

Choose the correct answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 34
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : $$NH_3$$ and $$NF_3$$ molecule have pyramidal shape with a lone pair of electrons on nitrogen atom. The resultant dipole moment of $$NH_3$$ is greater than that of $$NF_3$$. Reason (R) : In $$NH_3$$, the orbital dipole due to lone pair is in the same direction as the resultant dipole moment of the $$N-H$$ bonds. $$F$$ is the most electronegative element. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 35
Given below are two statements : Statement I : On passing $$HCl_{(g)}$$ through a saturated solution of $$BaCl_2$$, at room temperature white turbidity appears. Statement II : When HCl gas is passed through a saturated solution of NaCl, sodium chloride is precipitated due to common ion effect. In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 36
The correct statements from the following are : (A) The decreasing order of atomic radii of group 13 elements is $$Tl > In > Ga > Al > B$$. (B) Down the group 13 electronegativity decreases from top to bottom. (C) Al dissolves in dil. HCl and liberates $$H_2$$ but conc. $$HNO_3$$ renders Al passive by forming a protective oxide layer on the surface. (D) All elements of group 13 exhibits highly stable +1 oxidation state. (E) Hybridisation of Al in $$[Al(H_2O)_6]^{3+}$$ ion is $$sp^3d^2$$. Choose the correct answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 37
The correct nomenclature for the following compound is
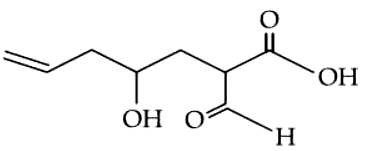
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 38
Match List I with List II :

Choose the correct answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 39
Identify $$A$$ and $$B$$ in the given chemical reaction sequence
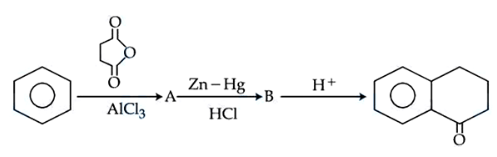
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 40
Consider the given chemical reaction :
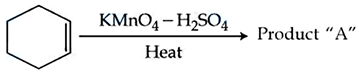
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 41
The quantity of silver deposited when one coulomb charge is passed through $$AgNO_3$$ solution :
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 42
For the electrochemical cell, if $$E^0_{(M^{2+}/M)} = 0.46$$ V and $$E^0_{(X/X^{2-})} = 0.34$$ V. Which of the following is correct?
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 43
The number of ions from the following that have the ability to liberate hydrogen from a dilute acid is ______. $$Ti^{2+}, Cr^{2+}$$ and $$V^{2+}$$
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 44
While preparing crystals of Mohr's salt, dil $$H_2SO_4$$ is added to a mixture of ferrous sulphate and ammonium sulphate, before dissolving this mixture in water, dil $$H_2SO_4$$ is added here to :
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 45
The number of complexes from the following with no electrons in the $$t_2$$ orbital is $$TiCl_4$$, $$[MnO_4]^-$$, $$[FeO_4]^{2-}$$, $$[FeCl_4]^-$$, $$[CoCl_4]^{2-}$$
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 46
The metal atom present in the complex MABXL (where A, B, X and L are unidentate ligands and M is metal) involves $$sp^3$$ hybridization. The number of geometrical isomers exhibited by the complex is:
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 47
Identify the major product in the following reaction.
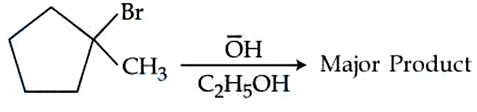
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 48
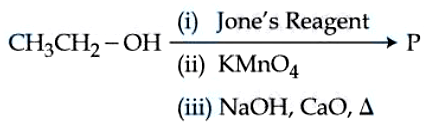
Consider the above reaction sequence and identify the major product $$P$$.
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 49
Which one of the following reactions is NOT possible?
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 50
Coagulation of egg, on heating is because of :
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 51
In an atom, total number of electrons having quantum numbers $$n = 4$$, $$|m_l| = 1$$ and $$m_s = -\frac{1}{2}$$ is ______
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 52
Number of compounds from the following with zero dipole moment is ______ $$HF$$, $$H_2$$, $$H_2S$$, $$CO_2$$, $$NH_3$$, $$BF_3$$, $$CH_4$$, $$CHCl_3$$, $$SiF_4$$, $$H_2O$$, $$BeF_2$$
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 53
Combustion of 1 mole of benzene is expressed at $$C_6H_6(l) + \frac{15}{2}O_2(g) \rightarrow 6CO_2(g) + 3H_2O(l)$$. The standard enthalpy of combustion of 2 mol of benzene is $$-'x'$$ kJ. $$x =$$ ______ Given: 1. Standard enthalpy of formation of 1 mol of $$C_6H_6(l)$$, for the reaction $$6C \text{(graphite)} + 3H_2(g) \rightarrow C_6H_6(l)$$ is $$48.5 \text{ kJ mol}^{-1}$$. 2. Standard enthalpy of formation of 1 mol of $$CO_2(g)$$, for the reaction $$C \text{(graphite)} + O_2(g) \rightarrow CO_2(g)$$ is $$-393.5 \text{ kJ mol}^{-1}$$. 3. Standard enthalpy of formation of 1 mol of $$H_2O(l)$$, for the reaction $$H_2(g) + \frac{1}{2}O_2(g) \rightarrow H_2O(l)$$ is $$-286 \text{ kJ mol}^{-1}$$.
789
456
123
0.-
Clear All
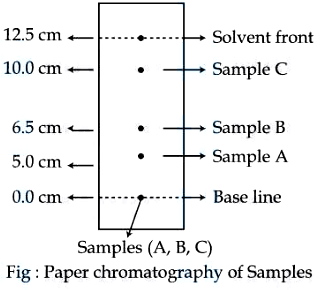
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 54
Using the given figure, the ratio of $$R_f$$ values of sample A and sample C is $$x \times 10^{-2}$$. Value of $$x$$ is _____
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 55
Considering acetic acid dissociates in water, its dissociation constant is $$6.25 \times 10^{-5}$$. If $$5$$ mL of acetic acid is dissolved in 1 litre water, the solution will freeze at $$-x \times 10^{-2} °C$$, provided pure water freezes at $$0°C$$. $$x =$$ ______ . (Nearest integer) Given : $$(K_f)_{water} = 1.86 \text{ K kg mol}^{-1}$$, density of acetic acid is $$1.2 \text{ g mL}^{-1}$$, molar mass of water $$= 18 \text{ g mol}^{-1}$$, molar mass of acetic acid $$= 60 \text{ g mol}^{-1}$$, density of water $$= 1 \text{ g cm}^{-3}$$. Acetic acid dissociates as $$CH_3COOH \rightleftharpoons CH_3COO^- + H^+$$
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 56
Consider the following single step reaction in gas phase at constant temperature. $$2A_{(g)} + B_{(g)} \rightarrow C_{(g)}$$. The initial rate of the reaction is recorded as $$r_1$$ when the reaction starts with $$1.5$$ atm pressure of A and $$0.7$$ atm pressure of B. After some time, the rate $$r_2$$ is recorded when the pressure of C becomes $$0.5$$ atm. The ratio $$r_1 : r_2$$ is ______ $$\times 10^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 57
The fusion of chromite ore with sodium carbonate in the presence of air leads to the formation of products A and B along with the evolution of $$CO_2$$. The sum of spin-only magnetic moment values of A and B is ______ B.M. (Nearest integer) [Given atomic number : C : 6, Na : 11, O : 8, Fe : 26, Cr : 24]
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 58
In the Claisen-Schmidt reaction to prepare $$351$$ g of dibenzalacetone using $$87$$ g of acetone, the amount of benzaldehyde required is ______ g. (Nearest integer)
789
456
123
0.-
Clear All
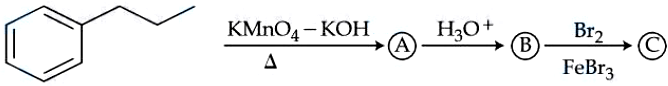
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 59
The product (C) in the following sequence of reactions has ______ $$\pi$$ bonds.
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Chemistry - Question 60
$$X$$ g of ethanamine was subjected to reaction with $$NaNO_2/HCl$$ followed by hydrolysis to liberate $$N_2$$ and HCl. The HCl generated was completely neutralised by 0.2 moles of NaOH. $$X$$ is ______ g.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




