Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The fusion of chromite ore with sodium carbonate in the presence of air leads to the formation of products A and B along with the evolution of $$CO_2$$. The sum of spin-only magnetic moment values of A and B is ______ B.M. (Nearest integer) [Given atomic number : C : 6, Na : 11, O : 8, Fe : 26, Cr : 24]
Correct Answer: 6
When chromite ore ($$FeCr_2O_4$$) is fused with $$Na_2CO_3$$ in the presence of air ($$O_2$$), the reaction is:
$$4FeCr_2O_4 + 8Na_2CO_3 + 7O_2 \rightarrow 8Na_2CrO_4 + 2Fe_2O_3 + 8CO_2$$
The products are sodium chromate ($$Na_2CrO_4$$) and ferric oxide ($$Fe_2O_3$$).
In $$Na_2CrO_4$$, chromium is in the +6 oxidation state, so $$Cr^{6+}$$ has the electronic configuration [Ar] $$3d^0$$ with zero unpaired electrons. The spin-only magnetic moment is given by $$\mu = \sqrt{n(n+2)}$$, which here becomes $$\mu = \sqrt{0(0+2)} = 0 \text{ B.M.}$$.
In $$Fe_2O_3$$, iron is in the +3 oxidation state, hence $$Fe^{3+}$$ has [Ar] $$3d^5$$ with five unpaired electrons. Applying the same formula yields $$\mu = \sqrt{5(5+2)} = \sqrt{35} \approx 5.92 \text{ B.M.}$$.
Therefore, the sum of the magnetic moments is $$\mu_A + \mu_B = 0 + 5.92 = 5.92 \approx 6 \text{ B.M.}$$, and the final answer is 6.
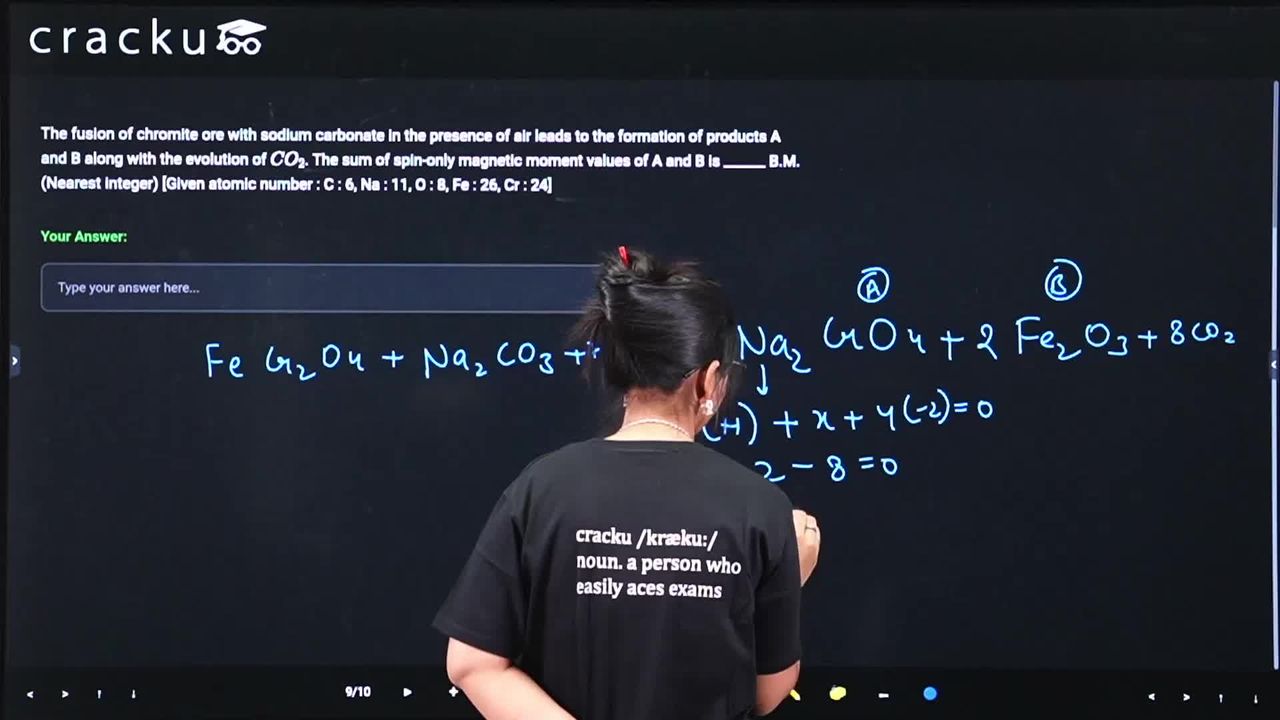
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
