Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
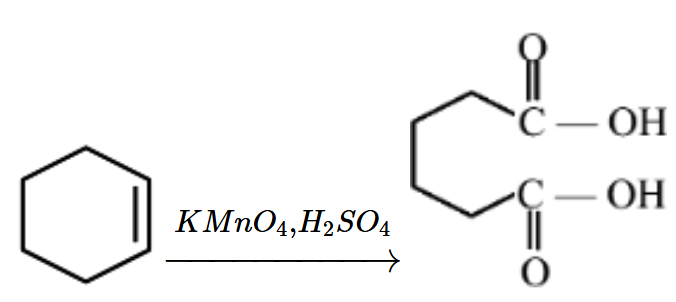
This reagent combination acts as a strong oxidizing agent that completely cleaves the carbon-carbon double bond ($$\text{C}=\text{C}$$).
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
