Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
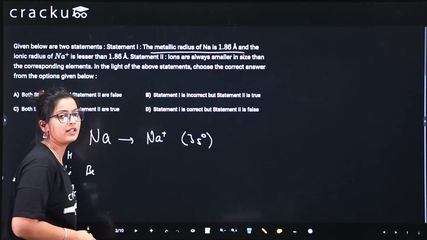
Given below are two statements : Statement I : The metallic radius of Na is $$1.86$$ Å and the ionic radius of $$Na^+$$ is lesser than $$1.86$$ Å. Statement II : Ions are always smaller in size than the corresponding elements. In the light of the above statements, choose the correct answer from the options given below :
Statement I: The metallic radius of Na is 1.86 A and the ionic radius of $$Na^+$$ is less than 1.86 A.
Statement II: Ions are always smaller in size than the corresponding elements.
Evaluation of Statement I:
When sodium loses an electron to form $$Na^+$$, it loses the entire outermost shell (3s). The resulting ion has the electronic configuration of neon, with a much smaller radius. The ionic radius of $$Na^+$$ is approximately 0.95 A, which is indeed less than 1.86 A. Statement I is correct.
Evaluation of Statement II:
This statement claims ions are always smaller than their parent atoms. This is true for cations (positive ions lose electrons, reducing electron-electron repulsion and the outermost shell). However, for anions (negative ions), the addition of electrons increases electron-electron repulsion and the ionic radius is larger than the atomic radius. For example, $$Cl^-$$ (1.81 A) is larger than Cl (0.99 A). Therefore, Statement II is false.
Statement I is correct but Statement II is false, which corresponds to Option 4.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
