NTA JEE Main 25th June 2022 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 31
The pair, in which ions are isoelectronic with $$Al^{3+}$$ is
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 32
Bonding in which of the following diatomic molecule(s) become(s) stronger, on the basis of MO Theory, by removal of an electron?
(A) NO
(B) $$N_2$$
(C) $$O_2$$
(D) $$C_2$$
(E) $$B_2$$
Choose the most appropriate answer from the options given below :
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 33
Number of electron deficient molecules among the following $$PH_3, B_2H_6, CCl_4, NH_3, LiH$$ and $$BCl_3$$ is
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 34
Which one of the following alkaline earth metal ions has the highest ionic mobility in its aqueous solution?
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 35
Phenol on reaction with dilute nitric acid, gives two products. Which method will be most efficient for large scale separation?
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 36
In the following structures, which one is having staggered conformation with maximum dihedral angle?
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 37
The IUPAC name of ethylidene chloride is
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 38
The product formed in the following reaction.
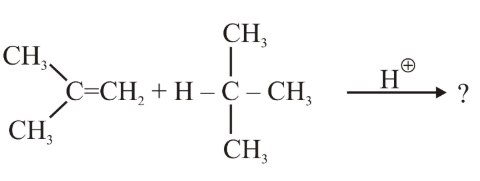
is:
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 39
The eutrophication of water body results in
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 40
Incorrect statement for Tyndall effect is
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 41
Leaching of gold with dilute aqueous solution of NaCN in presence of oxygen gives complex A, which on reaction with zinc forms the elemental gold and another complex B. A and B, respectively are
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 42
Cerium IV has a noble gas configuration. Which of the following is the correct statement about it?
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 43
Among the following, which is the strongest oxidizing agent?
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 44
White precipitate of AgCl dissolves in aqueous ammonia solution due to formation of
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 45
The major product in the reaction
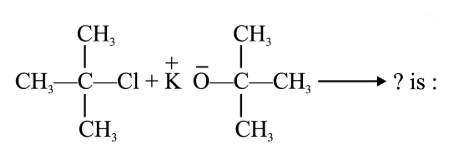
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 46
The intermediate X, in the reaction
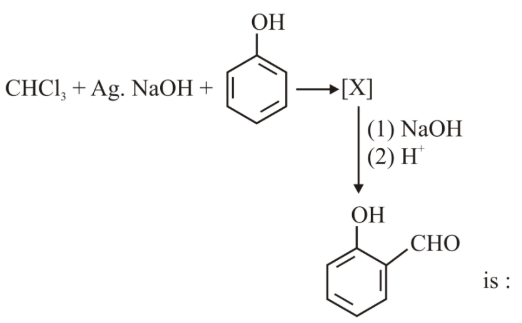
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 47
In the following reaction :
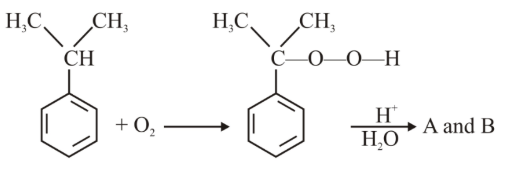
The compounds A and B respectively are
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 48
The reaction of
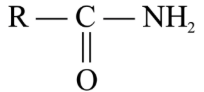
with bromine and KOH gives $$RNH_2$$ as the end product. Which one of the following is the intermediate product formed in this reaction?
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 49
Using very little soap while washing clothes, does not serve the purpose of cleaning of clothes, because
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 50
Which one of the following is an example of artificial sweetner?
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 51
The number of N atoms in $$681$$ g of $$C_7H_5N_3O_6$$ is $$x \times 10^{21}$$. The value of $$x$$ is ______ (Nearest Integer)
$$N_A = 6.02 \times 10^{23}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 52
$$1$$ L aqueous solution of $$H_2SO_4$$ contains $$0.02$$ m mol $$H_2SO_4$$. $$50\%$$ of this solution is diluted with deionized water to give $$1$$ L solution A. In solution A, $$0.01$$ m mol of $$H_2SO_4$$ are added. Total m mols of $$H_2SO_4$$ in the final solution is ______ $$\times 10^{-3}$$ m moles.
789
456
123
0.-
Clear All
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 53
Number of grams of bromine that will completely react with $$5.0$$ g of pent-1-ene is ______ $$\times 10^{-2}$$ g. (Atomic mass of Br $$= 80$$ g/mol) [Nearest integer]
789
456
123
0.-
Clear All
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 54
The longest wavelength of light that can be used for the ionisation of lithium ion $$Li^{2+}$$ is $$x \times 10^{-8}$$ m. The value of $$x$$ is ______ (Nearest Integer)
(Given : Energy of the electron in the first shell of the hydrogen atom is $$-2.2 \times 10^{-18}$$ J; $$h = 6.63 \times 10^{-34}$$ Js and $$c = 3 \times 10^{8}$$ ms$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 55
The standard entropy change for the reaction
$$4Fe(s) + 3O_2(g) \to 2Fe_2O_3(s)$$ is $$-550$$ J K$$^{-1}$$ at $$298$$ K
[Given : The standard enthalpy change for the reaction is $$-165$$ kJ mol$$^{-1}$$]. The temperature in K at which the reaction attains equilibrium is (Nearest Integer) ______
789
456
123
0.-
Clear All
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 56
The standard free energy change $$\Delta G°$$ for $$50\%$$ dissociation of $$N_2O_4$$ into $$NO_2$$ at $$27°$$C and $$1$$ atm pressure is $$-x$$ J mol$$^{-1}$$. The value of $$x$$ is ______ J. (Nearest Integer)
[Given : $$R = 8.31$$ J K$$^{-1}$$ mol$$^{-1}$$, $$\log 1.33 = 0.1239$$, $$\ln 10 = 2.3$$]
789
456
123
0.-
Clear All
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 57
The distance between $$Na^+$$ and $$Cl^-$$ ions in solid NaCl of density $$43.1$$ g cm$$^{-3}$$ is ______ $$\times 10^{-10}$$ m. (Nearest Integer)
(Given : $$N_A = 6.02 \times 10^{23}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 58
In a cell, the following reactions take place
$$Fe^{2+} \to Fe^{3+} + e^-$$ $$E^\circ_{Fe^{3+}/Fe^{2+}} = 0.77$$ V
$$2I^- \to I_2 + 2e^-$$ $$E^\circ_{I_2/I^-} = 0.54$$ V
The standard electrode potential for the spontaneous reaction in the cell is $$x \times 10^{-2}$$ V at $$298$$ K. The value of $$x$$ is ______ (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 59
For a given chemical reaction $$\gamma_1 A + \gamma_2 B \to \gamma_3 C + \gamma_4 D$$. Concentration of C changes from $$10$$ mmol dm$$^{-3}$$ to $$20$$ mmol dm$$^{-3}$$ in $$10$$ s. Rate of appearance of D is $$1.5$$ times the rate of disappearance of B which is twice the rate of disappearance of A. The rate of appearance of D has been experimentally determined to be $$9$$ mmol dm$$^{-3}$$ s$$^{-1}$$. Therefore the rate of reaction is ______ mmol dm$$^{-3}$$ s$$^{-1}$$. (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th June 2022 Shift 1 - Chemistry - Question 60
If $$[CuH_2O_4]^{2+}$$ absorbs a light of wavelength $$600$$ nm for d-d transition, then the value of octahedral crystal field splitting energy for $$[CuH_2O_6]^{2+}$$ will be ______ $$\times 10^{-21}$$ J [Nearest integer]
(Given : $$h = 6.63 \times 10^{-34}$$ Js and $$c = 3.08 \times 10^{8}$$ ms$$^{-1}$$)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




