Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The number of N atoms in $$681$$ g of $$C_7H_5N_3O_6$$ is $$x \times 10^{21}$$. The value of $$x$$ is ______ (Nearest Integer)
$$N_A = 6.02 \times 10^{23}$$ mol$$^{-1}$$
Correct Answer: 5418
We need to find the number of nitrogen atoms in 681 g of $$C_7H_5N_3O_6$$.
The molar mass of $$C_7H_5N_3O_6$$ is determined by $$M = 7(12) + 5(1) + 3(14) + 6(16) = 84 + 5 + 42 + 96 = 227 \text{ g/mol}$$, so the number of moles in 681 g is $$n = \frac{681}{227} = 3 \text{ mol}$$.
Each molecule contains 3 nitrogen atoms, hence the total number of nitrogen atoms is $$\text{Total N atoms} = 3 \text{ mol} \times 3 \times 6.02 \times 10^{23} \text{ mol}^{-1} = 9 \times 6.02 \times 10^{23} = 54.18 \times 10^{23} = 5418 \times 10^{21}$$.
Expressing this as $$x \times 10^{21}$$ yields $$x = 5418$$, so the correct answer is 5418.
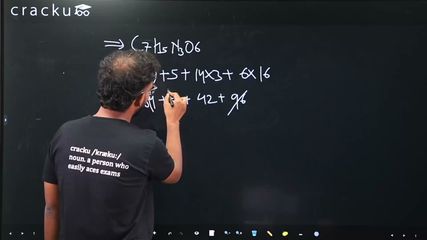
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
