Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Leaching of gold with dilute aqueous solution of NaCN in presence of oxygen gives complex A, which on reaction with zinc forms the elemental gold and another complex B. A and B, respectively are
This question is about the MacArthur-Forrest cyanide process for gold extraction.
Leaching reaction (formation of complex A): Gold reacts with dilute NaCN solution in the presence of oxygen:
$$4Au(s) + 8NaCN(aq) + O_2(g) + 2H_2O(l) \rightarrow 4Na[Au(CN)_2](aq) + 4NaOH(aq)$$
The gold complex formed is $$[Au(CN)_2]^-$$ (dicyanoaurate(I) ion). So complex A = $$[Au(CN)_2]^-$$.
Recovery with zinc (formation of complex B): Zinc displaces gold from the cyanide complex:
$$2[Au(CN)_2]^-(aq) + Zn(s) \rightarrow 2Au(s) + [Zn(CN)_4]^{2-}(aq)$$
The zinc complex formed is $$[Zn(CN)_4]^{2-}$$ (tetracyanozincate(II) ion). So complex B = $$[Zn(CN)_4]^{2-}$$.
Match with options: Option A: $$[Au(CN)_2]^-$$ and $$[Zn(CN)_4]^{2-}$$ — Matches both A and B. Correct.
Option B: $$[Au(CN)_4]^-$$ and $$[Zn(CN)_2(OH)_2]^{2+}$$ — Incorrect coordination numbers and charges.
Option C: $$[Au(CN)_2]^-$$ and $$[Zn(OH)_4]^{2-}$$ — Complex B is wrong.
Option D: $$[Au(CN)_4]^{2-}$$ and $$[Zn(CN)_6]^{4-}$$ — Both are incorrect.
The correct answer is Option A: $$[Au(CN)_2]^-$$ and $$[Zn(CN)_4]^{2-}$$.
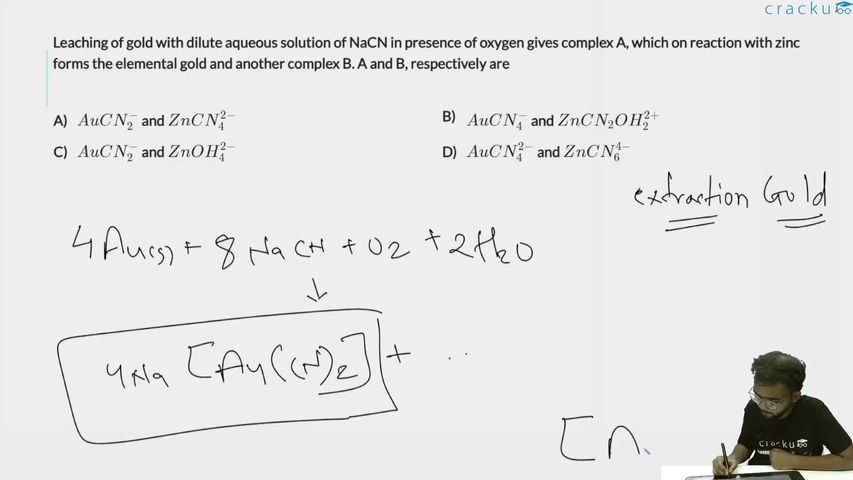
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
