Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
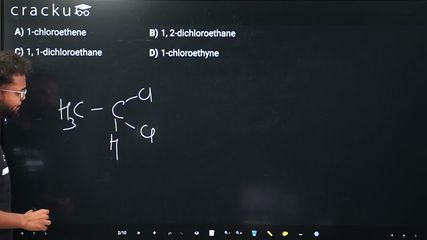
Ethylidene chloride is a common name for a compound where two chlorine atoms are attached to the same carbon of ethane.
Identify the structure of ethylidene chloride: The prefix "ethylidene" refers to the $$CH_3CH=$$ group (a divalent group derived from ethane by removing two hydrogens from the same carbon). When two chlorine atoms attach to this group, we get:
$$CH_3CHCl_2$$
Apply IUPAC nomenclature: The parent chain is ethane (2 carbons). Both chlorine atoms are on carbon-1 (the carbon bearing the most substituents when numbering to give the lowest locants).
$$\Rightarrow$$ 1, 1-dichloroethane
Evaluate the options: Option A: 1-chloroethene — This would be vinyl chloride ($$CH_2=CHCl$$), which is incorrect.
Option B: 1, 2-dichloroethane — This is ethylene dichloride ($$ClCH_2CH_2Cl$$), where chlorines are on different carbons. Incorrect.
Option C: 1, 1-dichloroethane — This matches $$CH_3CHCl_2$$. Correct.
Option D: 1-chloroethyne — This would be $$HC \equiv CCl$$, which is incorrect.
The correct answer is Option C: 1, 1-dichloroethane.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
