Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
To find the pair of ions isoelectronic with $$Al^{3+}$$, note that aluminium (Al) has atomic number 13, so $$Al^{3+}$$ has $$13 - 3 = 10$$ electrons.
In Option A, $$Br^-$$ has $$35 + 1 = 36$$ electrons and $$Be^{2+}$$ has $$4 - 2 = 2$$ electrons, so neither has 10. In Option B, $$Cl^-$$ has $$17 + 1 = 18$$ electrons and $$Li^+$$ has $$3 - 1 = 2$$ electrons, so neither has 10. In Option C, $$S^{2-}$$ has $$16 + 2 = 18$$ electrons and $$K^+$$ has $$19 - 1 = 18$$ electrons, so neither has 10.
In Option D, $$O^{2-}$$ has $$8 + 2 = 10$$ electrons and $$Mg^{2+}$$ has $$12 - 2 = 10$$ electrons, so both have 10 electrons.
Since both $$O^{2-}$$ and $$Mg^{2+}$$ have the same number of electrons as $$Al^{3+}$$, they are isoelectronic. The correct answer is Option D.
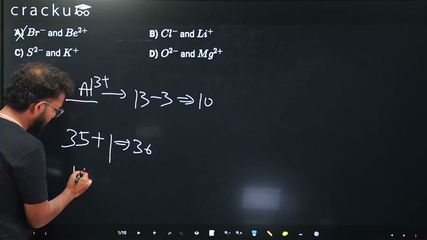
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
