Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
In a cell, the following reactions take place
$$Fe^{2+} \to Fe^{3+} + e^-$$ $$E^\circ_{Fe^{3+}/Fe^{2+}} = 0.77$$ V
$$2I^- \to I_2 + 2e^-$$ $$E^\circ_{I_2/I^-} = 0.54$$ V
The standard electrode potential for the spontaneous reaction in the cell is $$x \times 10^{-2}$$ V at $$298$$ K. The value of $$x$$ is ______ (Nearest Integer)
Correct Answer: 23
We need to find the standard electrode potential for the spontaneous cell reaction. First, we identify the half-reactions and their standard reduction potentials: $$Fe^{3+} + e^- \to Fe^{2+}$$, $$E°= 0.77$$ V and $$I_2 + 2e^- \to 2I^-$$, $$E° = 0.54$$ V.
For a spontaneous reaction, the species with the higher reduction potential acts as the cathode and is reduced, whereas the one with the lower reduction potential acts as the anode and is oxidized. Therefore, the cathode (reduction) is $$Fe^{3+} + e^- \to Fe^{2+}$$, $$E° = 0.77$$ V and the anode (oxidation) is $$2I^- \to I_2 + 2e^-$$, $$E° = 0.54$$ V.
Then, the standard cell potential is calculated by subtracting the anode potential from the cathode potential: $$E°_{cell} = E°_{cathode} - E°_{anode} = 0.77 - 0.54 = 0.23 \text{ V}$$
Finally, we express this in the required form as $$E°_{cell} = 0.23$$ V $$= 23 \times 10^{-2}$$ V. Consequently, the correct answer is $$x = \mathbf{23}$$.
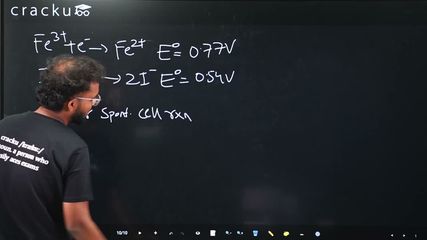
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
