Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
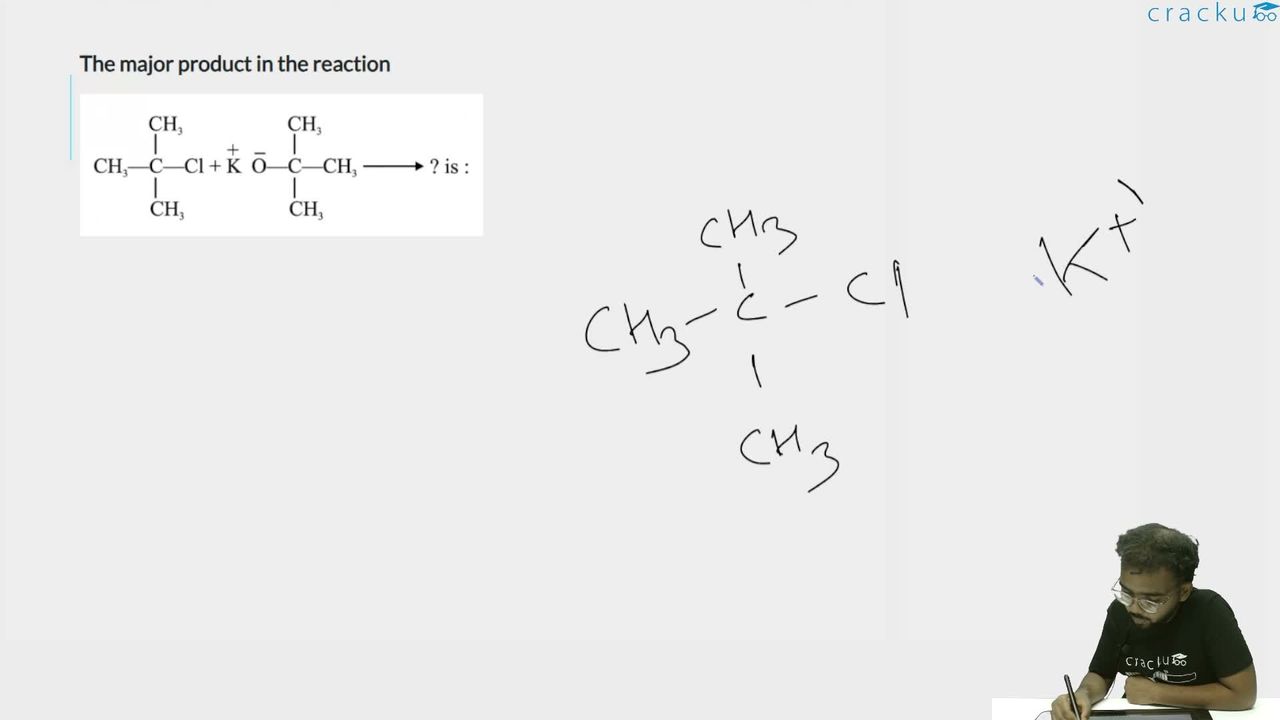
We need to find the major product when tert-butyl chloride reacts with potassium tert-butoxide.
Identify the substrate and reagent: Substrate: $$(CH_3)_3C{-}Cl$$ (tert-butyl chloride) — a tertiary alkyl halide.
Reagent: $$K^+ \; ^-OC(CH_3)_3$$ (potassium tert-butoxide) — a strong, bulky base.
Determine the reaction pathway: With a tertiary substrate:
$$\bullet$$ $$S_N2$$ is not possible due to steric hindrance at the tertiary carbon.
$$\bullet$$ $$S_N1$$ could occur but is disfavored because tert-butoxide is a strong base, not just a nucleophile.
$$\bullet$$ The bulky tert-butoxide base strongly favors E2 elimination over substitution.
Determine the elimination product: E2 elimination of HCl from $$(CH_3)_3CCl$$:
$$(CH_3)_3C{-}Cl \xrightarrow{KOC(CH_3)_3} (CH_3)_2C{=}CH_2 + KCl$$
The product is 2-methylprop-1-ene (isobutylene), formed by removal of H from one of the methyl groups and Cl from the tertiary carbon.
Evaluate the options: Option A: t-Butyl ethyl ether — Would require $$S_N$$ reaction, which is not favored here.
Option B: 2-Methyl pent-1-ene — Incorrect carbon count; the substrate has only 4 carbons.
Option C: 2, 2-Dimethyl butane — Would require C-C bond formation, not possible in this reaction.
Option D: 2-Methyl prop-1-ene — Correct. This is the E2 elimination product.
The correct answer is Option D: 2-Methyl prop-1-ene.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
