Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Number of grams of bromine that will completely react with $$5.0$$ g of pent-1-ene is ______ $$\times 10^{-2}$$ g. (Atomic mass of Br $$= 80$$ g/mol) [Nearest integer]
Correct Answer: 1143
To determine the mass of bromine that reacts completely with 5.0 g of pent-1-ene, we first consider the addition reaction of pent-1-ene ($$CH_2{=}CHCH_2CH_2CH_3$$) with bromine across the double bond:
$$CH_2{=}CHCH_2CH_2CH_3 + Br_2 \rightarrow CH_2BrCHBrCH_2CH_2CH_3$$ Because one mole of pent-1-ene reacts with one mole of $$Br_2$$, the stoichiometric ratio is 1:1.
Next, the molar mass of pent-1-ene is calculated from its molecular formula $$C_5H_{10}$$:
$$M = 5(12) + 10(1) = 60 + 10 = 70 \text{ g/mol}$$ As a result, the number of moles in 5.0 g of pent-1-ene is:
$$n = \frac{5.0}{70} = \frac{1}{14} \text{ mol}$$.
Since the molar ratio between pent-1-ene and $$Br_2$$ is 1:1, the required amount of $$Br_2$$ is also $$\frac{1}{14}$$ mol. Given that the molar mass of $$Br_2$$ is 2 × 80 = 160 g/mol, the mass of $$Br_2$$ needed becomes:
$$\text{Mass of } Br_2 = \frac{1}{14} \times 160 = \frac{160}{14} = 11.4286 \text{ g}$$.
Finally, expressing this mass in the desired format gives:
$$11.4286 \text{ g} = 1142.86 \times 10^{-2} \text{ g} \approx 1143 \times 10^{-2} \text{ g}$$ therefore the correct answer is 1143.
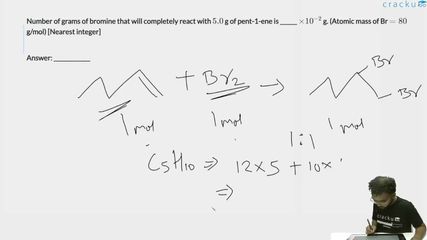
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
