Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
We need to identify the strongest oxidizing agent among $$Mn^{3+}$$, $$Ti^{3+}$$, $$Fe^{3+}$$, and $$Cr^{3+}$$.
Understand what makes a strong oxidizing agent: A strong oxidizing agent has a high tendency to gain electrons (get reduced). This corresponds to a high standard reduction potential for the $$M^{3+}/M^{2+}$$ couple.
Analyze the electronic configurations: $$\bullet$$ $$Mn^{3+}$$: $$[Ar] 3d^4$$. On gaining an electron, it becomes $$Mn^{2+}$$: $$[Ar] 3d^5$$ (half-filled, extra stable).
$$\bullet$$ $$Ti^{3+}$$: $$[Ar] 3d^1$$. On gaining an electron, $$Ti^{2+}$$: $$[Ar] 3d^2$$ (no special stability).
$$\bullet$$ $$Fe^{3+}$$: $$[Ar] 3d^5$$ (already half-filled, stable). On gaining an electron, $$Fe^{2+}$$: $$[Ar] 3d^6$$ (no special stability).
$$\bullet$$ $$Cr^{3+}$$: $$[Ar] 3d^3$$ (half-filled $$t_{2g}$$, stable). On gaining an electron, $$Cr^{2+}$$: $$[Ar] 3d^4$$ (no special stability).
Compare reduction potentials: The standard reduction potentials ($$E^\circ$$ for $$M^{3+}/M^{2+}$$) are:
$$\bullet$$ $$Mn^{3+}/Mn^{2+}$$: $$E^\circ = +1.51$$ V (very high, due to extra stability of half-filled $$d^5$$)
$$\bullet$$ $$Fe^{3+}/Fe^{2+}$$: $$E^\circ = +0.77$$ V
$$\bullet$$ $$Cr^{3+}/Cr^{2+}$$: $$E^\circ = -0.41$$ V
$$\bullet$$ $$Ti^{3+}/Ti^{2+}$$: $$E^\circ = -0.37$$ V
$$Mn^{3+}$$ has the highest reduction potential because its reduction product $$Mn^{2+}$$ ($$d^5$$) has the extra stability associated with the half-filled d subshell.
The correct answer is Option A: $$Mn^{3+}$$.
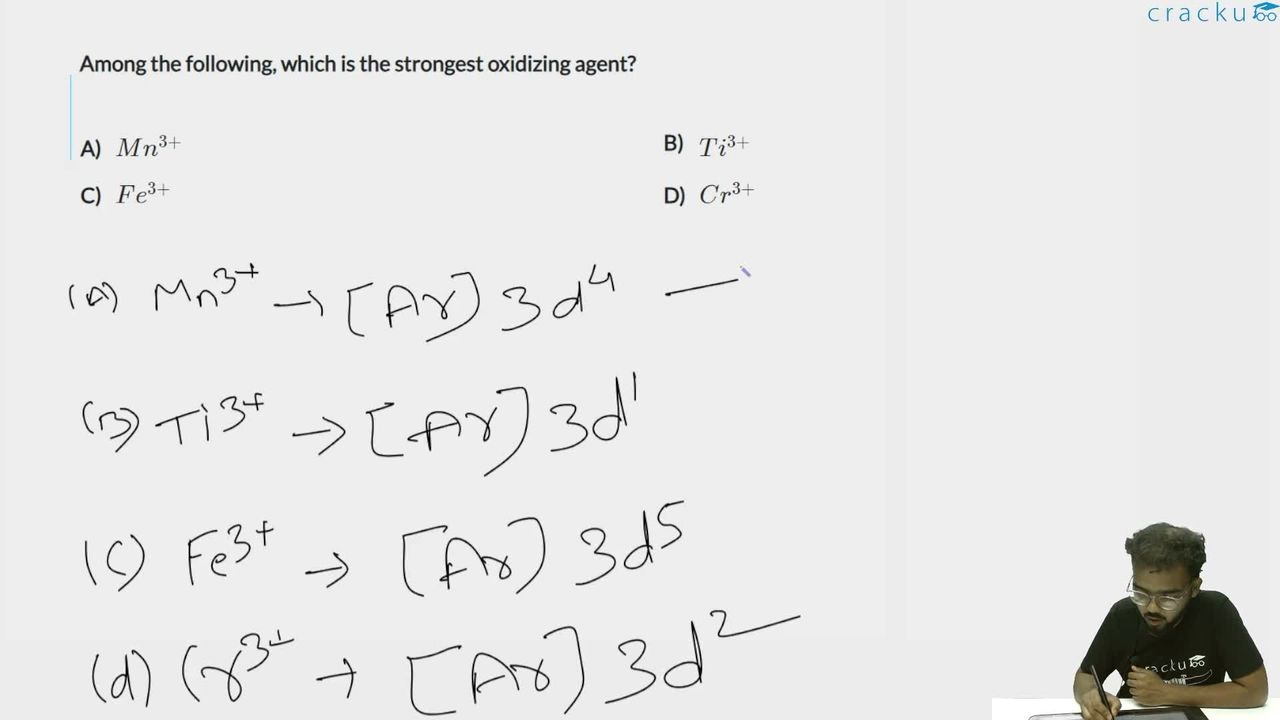
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
