NTA JEE Mains 6th April 2024 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 31
The density of 'x' M solution ('X' molar) of NaOH is $$1.12 \text{ g mL}^{-1}$$, while in molality, the concentration of the solution is $$3 \text{ m (3 molal)}$$. Then $$x$$ is (Given : Molar mass of NaOH is $$40 \text{ g/mol}$$)
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 32
The electron affinity values are negative for A. $$\text{Be} \rightarrow \text{Be}^-$$ B. $$\text{N} \rightarrow \text{N}^-$$ C. $$\text{O} \rightarrow \text{O}^{2-}$$ D. $$\text{Na} \rightarrow \text{Na}^-$$ E. $$\text{Al} \rightarrow \text{Al}^-$$. Choose the most appropriate answer from the options given below :
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 33
Which of the following material is not a semiconductor.
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 34
Match List I with List II
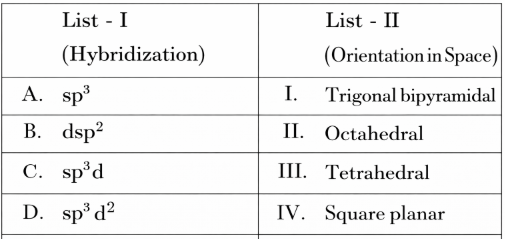
Choose the correct answer from the options given below:
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 35
Match List I with List II
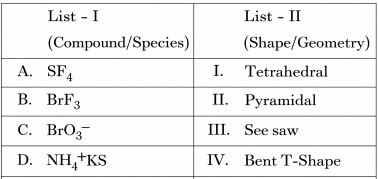
Choose the correct answer from the options given below:
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 36
Match List I with List II
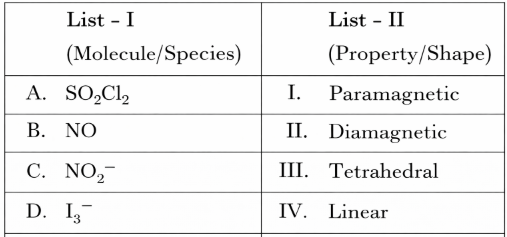
Choose the correct answer from the options given below:
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 37
At $$-20°C$$ and 1 atm pressure, a cylinder is filled with equal number of $$H_2$$, $$I_2$$ and $$HI$$ molecules for the reaction $$H_2(g) + I_2(g) \rightleftharpoons 2HI(g)$$, the $$K_p$$ for the process is $$x \times 10^{-1}$$. $$x =$$ _______ [Given : $$R = 0.082 \text{ L atm K}^{-1} \text{mol}^{-1}$$]
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 38
Functional group present in sulphonic acids is :
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 39
Which of the following statements are correct? A. Glycerol is purified by vacuum distillation because it decomposes at its normal boiling point. B. Aniline can be purified by steam distillation as aniline is miscible in water. C. Ethanol can be separated from ethanol water mixture by azeotropic distillation because it forms azeotrope. D. An organic compound is pure, if mixed M.P. is remained same. Choose the most appropriate answer from the options given below :
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 40
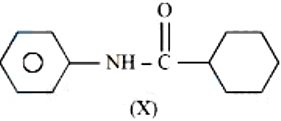
Which of the following is metamer of the given compound (X)?
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 41
Given below are two statements: Statement I : Gallium is used in the manufacturing of thermometers. Statement II : A thermometer containing gallium is useful for measuring the freezing point of brine solution (256 K). In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 42
A conductivity cell with two electrodes (dark side) are half filled with infinitely dilute aqueous solution of a weak electrolyte. If volume is doubled by adding more water at constant temperature, the molar conductivity of the cell will -
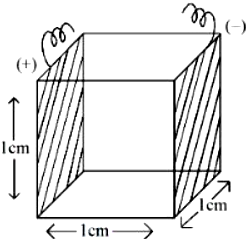
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 43
The number of elements from the following that do not belong to lanthanoids is $$Eu, Cm, Er, Tb, Yb$$ and $$Lu$$
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 44
Match List I with List II
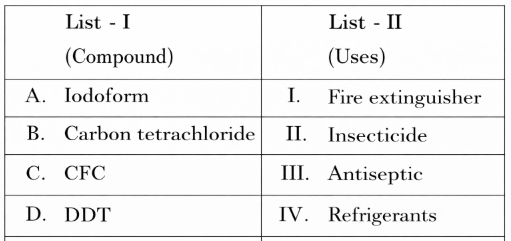
Choose the correct answer from the options given below:
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 45
The following complexes $$[CoCl(NH_3)_5]^{2+}$$ (A), $$[Co(CN)_6]^{3-}$$ (B), $$[Co(NH_3)_5(H_2O)]^{3+}$$ (C), $$[Cu(H_2O)_4]^{2+}$$ (D). The correct order of A, B, C and D in terms of wavenumber of light absorbed is :
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 46
Given below are two statements : Statement I : Picric acid is 2,4,6 - trinitrotoluene. Statement II : Phenol - 2,4 - disulphonic acid is treated with Conc. $$HNO_3$$ to get picric acid. In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 47
In Reimer - Tiemann reaction, phenol is converted into salicylaldehyde through an intermediate. The structure of intermediate is _____
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 48
Which among the following aldehydes is most reactive towards nucleophilic addition reactions?
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 49
Match List I with List II
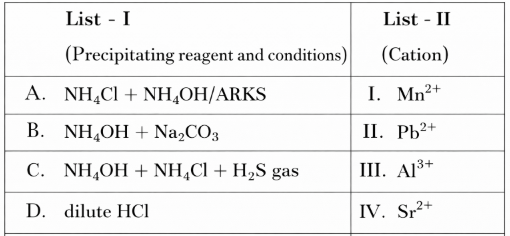
Choose the correct answer from the options given below:
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 50
DNA molecule contains 4 bases whose structures are shown below. One of the structures is not correct, identify the incorrect base structure.
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 51
Frequency of the de-Broglie wave of electron in Bohr's first orbit of hydrogen atom is _______ $$\times 10^{13}$$ Hz (nearest integer). [Given : $$R_H$$ (Rydberg constant) $$= 2.18 \times 10^{-18}$$ J, $$h$$ (Planck's constant) $$= 6.6 \times 10^{-34}$$ J.s.]
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 52
Number of molecules from the following which can exhibit hydrogen bonding is _______ (nearest integer):
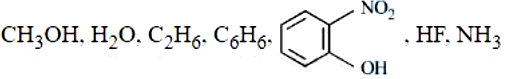
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 53
An ideal gas, $$\bar{C}_v = \frac{5}{2}R$$, is expanded adiabatically against a constant pressure of 1 atm until it doubles in volume. If the initial temperature and pressure is $$298 \text{ K}$$ and $$5 \text{ atm}$$, respectively then the final temperature is _______ K (nearest integer). [$$\bar{C}_v$$ is the molar heat capacity at constant volume]
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 54
The major product of the following reaction is $$P$$. $$CH_3C \equiv C-CH_3 \xrightarrow[\text{(ii) dil. KMnO}_4, 273\text{ K}]{\text{(i) Na/liq. NH}_3} P$$. Number of oxygen atoms present in product '$$P$$' is _______ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 55
Consider the dissociation of the weak acid HX as given below $$HX(aq) \rightleftharpoons H^+(aq) + X^-(aq)$$, $$K_a = 1.2 \times 10^{-5}$$ [$$K_a$$ : dissociation constant]. The osmotic pressure of $$0.03$$ M aqueous solution of HX at $$300 \text{ K}$$ is _______ $$\times 10^{-2}$$ bar (nearest integer). [Given : $$R = 0.083 \text{ L bar mol}^{-1} \text{K}^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 56
Time required for 99.9% completion of a first order reaction is _______ times the time required for completion of 90% reaction. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 57
Among $$CrO$$, $$Cr_2O_3$$ and $$CrO_3$$, the sum of spin-only magnetic moment values of basic and amphoteric oxides is _______ $$\times 10^{-2}$$ BM (nearest integer). (Given atomic number of Cr is 24)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 58
The difference in the 'spin-only' magnetic moment values of $$KMnO_4$$ and the manganese product formed during titration of $$KMnO_4$$ against oxalic acid in acidic medium is _______ BM. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 59
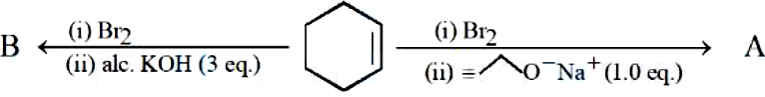
The major products from the following reaction sequence are product A and product B. The total sum of $$\pi$$ electrons in product A and product B are _______ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Chemistry - Question 60
$$9.3 \text{ g}$$ of pure aniline upon diazotisation followed by coupling with phenol gives an orange dye. The mass of orange dye produced (assume 100% yield/conversion) is _______ g. (nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




