Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
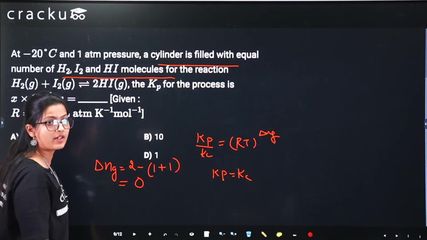
At $$-20°C$$ and 1 atm pressure, a cylinder is filled with equal number of $$H_2$$, $$I_2$$ and $$HI$$ molecules for the reaction $$H_2(g) + I_2(g) \rightleftharpoons 2HI(g)$$, the $$K_p$$ for the process is $$x \times 10^{-1}$$. $$x =$$ _______ [Given : $$R = 0.082 \text{ L atm K}^{-1} \text{mol}^{-1}$$]
The reaction is $$H_2(g) + I_2(g) \rightleftharpoons 2HI(g)$$.
The cylinder is filled with equal number of molecules of $$H_2$$, $$I_2$$, and $$HI$$ at $$-20°C$$ (253 K) and 1 atm.
Since the number of moles of gaseous reactants equals the number of moles of gaseous products ($$\Delta n_g = 0$$), $$K_p = K_c$$ for this reaction.
Let the number of moles of each gas be $$n$$. The partial pressures are all equal (since equal moles at same T, V): each gas has partial pressure $$P/3$$ where $$P = 1$$ atm.
$$K_p = \frac{(P_{HI})^2}{P_{H_2} \cdot P_{I_2}} = \frac{(P/3)^2}{(P/3)(P/3)} = \frac{P^2/9}{P^2/9} = 1$$
So $$K_p = 1 = x \times 10^{-1}$$.
This gives $$x = 10$$.
The correct answer is Option B: 10.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
