Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
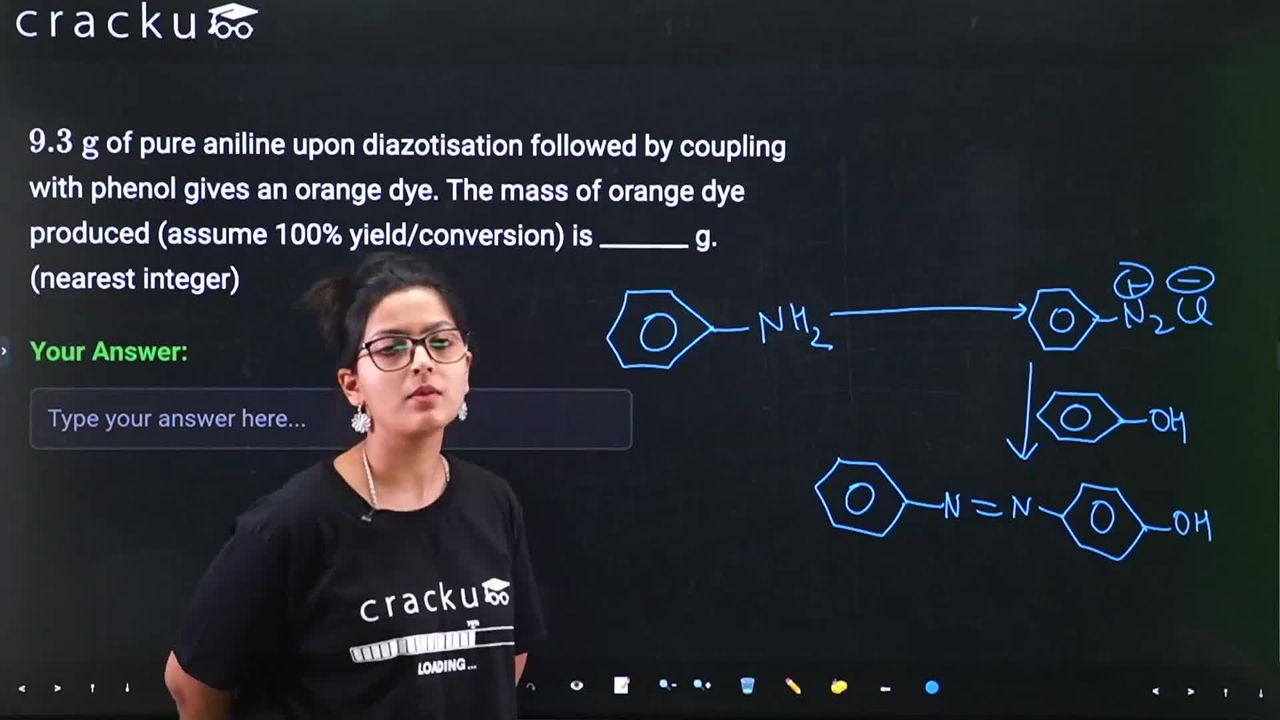
$$9.3 \text{ g}$$ of pure aniline upon diazotisation followed by coupling with phenol gives an orange dye. The mass of orange dye produced (assume 100% yield/conversion) is _______ g. (nearest integer)
Correct Answer: 20
We have 9.3 g of pure aniline which undergoes diazotisation followed by coupling with phenol to give an orange dye.
In the diazotisation step aniline reacts with nitrous acid (generated in situ from $$NaNO_2 + HCl$$) at 0-5 °C to form benzenediazonium chloride:
$$C_6H_5NH_2 + NaNO_2 + 2HCl \rightarrow C_6H_5N_2^+Cl^- + NaCl + 2H_2O$$
In the coupling step the diazonium salt couples with phenol in a weakly basic medium to produce p-hydroxyazobenzene (an orange azo dye):
$$C_6H_5N_2^+Cl^- + C_6H_5OH \rightarrow C_6H_5-N=N-C_6H_4-OH + HCl$$
To calculate the moles of aniline, we use its molar mass: $$6(12) + 5(1) + 14 + 2(1) = 93\text{ g/mol}$$, which gives $$\text{Moles of aniline} = \frac{9.3}{93} = 0.1\text{ mol}$$.
Since the stoichiometry is 1:1, one mole of aniline yields one mole of dye. The molar mass of p-hydroxyazobenzene ($$C_{12}H_{10}N_2O$$) is $$12(12) + 10(1) + 2(14) + 16 = 198\text{ g/mol}$$. Thus, the mass of dye produced is $$0.1 \times 198 = 19.8 \approx 20\text{ g}$$. Therefore, 20 g of the orange dye is obtained (to the nearest integer).
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
