Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
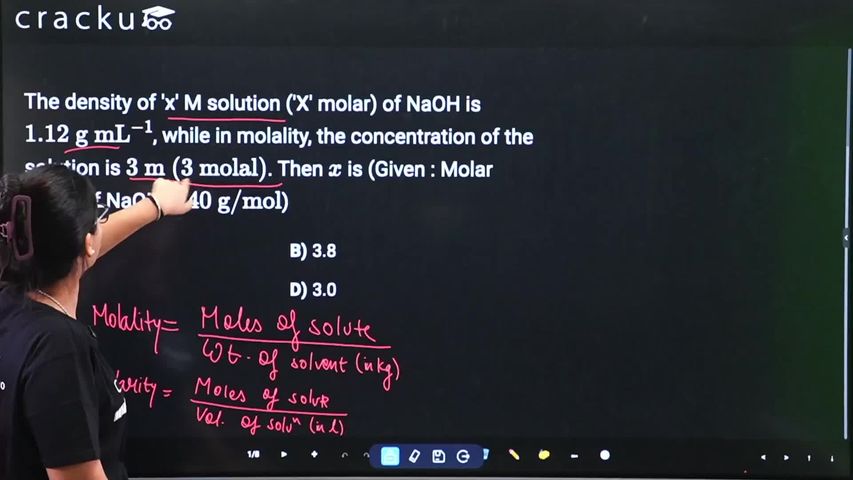
The density of 'x' M solution ('X' molar) of NaOH is $$1.12 \text{ g mL}^{-1}$$, while in molality, the concentration of the solution is $$3 \text{ m (3 molal)}$$. Then $$x$$ is (Given : Molar mass of NaOH is $$40 \text{ g/mol}$$)
Molality = 3 m means 3 mol NaOH per 1 kg solvent. Mass of NaOH = 3 × 40 = 120 g. Total mass = 1000 + 120 = 1120 g.
Volume = 1120/1.12 = 1000 mL = 1 L. Molarity = 3/1 = 3 M.
The correct answer is Option (4): 3.0.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
