Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The major product of the following reaction is $$P$$. $$CH_3C \equiv C-CH_3 \xrightarrow[\text{(ii) dil. KMnO}_4, 273\text{ K}]{\text{(i) Na/liq. NH}_3} P$$. Number of oxygen atoms present in product '$$P$$' is _______ (nearest integer)
Correct Answer: 2
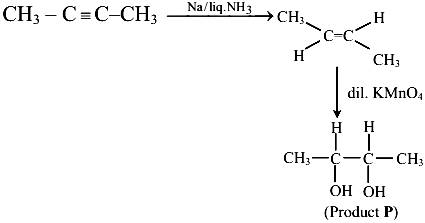
CH₃C≡C-CH₃ with Na/liq. NH₃ gives trans-2-butene (Birch reduction gives trans alkene).
Then dil. KMnO₄ at 273 K (cold) gives a diol: trans-but-2-ene-2,3-diol (syn addition giving glycol).
The product has 2 oxygen atoms (two OH groups).
The answer is 2.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
