NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 31
Given below are two statements:
Statement-I: The orbitals having same energy are called as degenerate orbitals.
Statement-II: In hydrogen atom, $$3p$$ and $$3d$$ orbitals are not degenerate orbitals.
In the light of the above statements, choose the most appropriate answer from the options given
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 32
Given below are the two statements: one is labeled as Assertion (A) and the other is labeled as Reason (R).
Assertion (A): There is a considerable increase in covalent radius from $$N$$ to $$P$$. However from $$As$$ to $$Bi$$ only a small increase in covalent radius is observed.
Reason (R): covalent and ionic radii in a particular oxidation state increases down the group.
In the light of the above statement, choose the most appropriate answer from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 33
Match List - I with List-II

NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 34
Structure of $$4-\text{Methylpent}-2-\text{enal}$$ is:
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 35
Example of vinylic halide is
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 36
Given below are two statements one is labeled as Assertion (A) and the other is labeled as Reason (R).
Assertion (A): $$CH_2=CH-CH_2-Cl$$ is an example of allyl halide
Reason (R): Allyl halides are the compounds in which the halogen atom is attached to $$sp^2$$ hybridised carbon atom.
In the light of the two above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 37
Which of the following molecule/species is most stable?
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 38
Compound $$A$$ formed in the following reaction reacts with $$B$$ gives the product $$C$$. Find out $$A$$ and $$B$$.
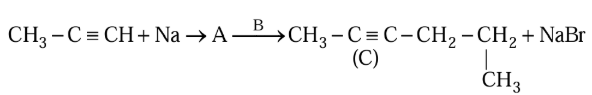
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 39
In the given reactions identify the reagent $$A$$ and reagent $$B$$
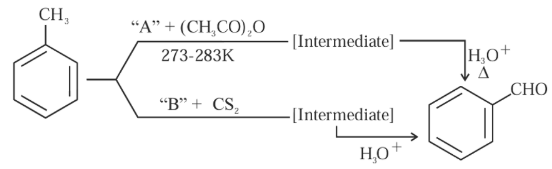
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 40
What happens to freezing point of benzene when small quantity of naphthalene is added to benzene?
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 41
Diamagnetic Lanthanoid ions are:
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 42
Match List-I with List-II

NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 43
Choose the correct Statements from the following:
(A) Ethane-1,2-diamine is a chelating ligand.
(B) Metallic aluminium is produced by electrolysis of aluminium oxide in presence of cryolite.
(C) Cyanide ion is used as ligand for leaching of silver.
(D) Phosphine acts as a ligand in Wilkinson catalyst.
(E) The stability constants of $$Ca^{2+}$$ and $$Mg^{2+}$$ are similar with EDTA complexes.
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 44
Aluminium chloride in acidified aqueous solution forms an ion having geometry
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 45
This reduction reaction is known as:
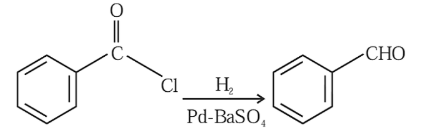
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 46
Following is a confirmatory test for aromatic primary amines. Identify reagent (A) and (B)
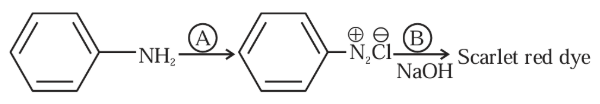
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 47
The final product $$A$$, formed in the following multistep reaction sequence is:
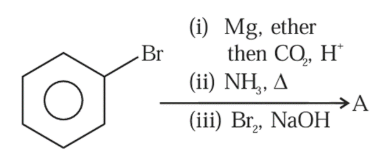
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 48
Given below are two statements:
Statement-I: The gas liberated on warming a salt with dil $$H_2SO_4$$, turns a piece of paper dipped in lead acetate into black, it is a confirmatory test for sulphide ion.
Statement-II: In statement-I the colour of paper turns black because of formation of lead sulphite.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 49
The Lassaigne's extract is boiled with dil $$HNO_3$$ before testing for halogens because,
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 50
Sugar which does not give reddish brown precipitate with Fehling's reagent is:
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 51
$$0.05 \text{ cm}$$ thick coating of silver is deposited on a plate of area $$0.05 \text{ m}^2$$. The number of silver atoms deposited on plate are _______ $$\times 10^{23}$$. (At mass $$Ag = 108$$, $$d = 7.9 \text{ g cm}^{-3}$$) Round off to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 52
If IUPAC name of an element is "Unununnium" then the element belongs to $$n$$th group of periodic table. The value of $$n$$ is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 53
The total number of molecular orbitals formed from $$2s$$ and $$2p$$ atomic orbitals of a diatomic molecule is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 54
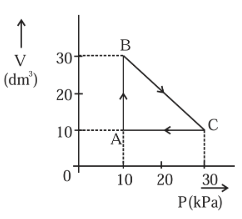
An ideal gas undergoes a cyclic transformation starting from the point $$A$$ and coming back to the same point by tracing the path $$A \rightarrow B \rightarrow C \rightarrow A$$ as shown in the diagram. The total work done in the process is _____ J.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 55
The pH at which $$Mg(OH)_2 [K_{sp} = 1 \times 10^{-11}]$$ begins to precipitate from a solution containing $$0.10 \text{ M } Mg^{2+}$$ ions is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 56
$$2MnO_4^- + bI^- + cH_2O \rightarrow xI_2 + yMnO_2 + zOH^-$$
If the above equation is balanced with integer coefficients, the value of $$z$$ is ________.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 57
On a thin layer chromatographic plate, an organic compound moved by $$3.5 \text{ cm}$$, while the solvent moved by $$5 \text{ cm}$$. The retardation factor of the organic compound is ____________ $$\times 10^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 58
The mass of sodium acetate $$(CH_3COONa)$$ required to prepare $$250 \text{ mL}$$ of $$0.35 \text{ M}$$ aqueous solution is _____ g. (Molar mass of $$CH_3COONa$$ is $$82.02 \text{ g mol}^{-1}$$) Round off to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 59
The rate of first order reaction is $$0.04 \text{ mol L}^{-1} \text{s}^{-1}$$ at $$10$$ minutes and $$0.03 \text{ mol L}^{-1} \text{s}^{-1}$$ at $$20$$ minutes after initiation. Half life of the reaction is ______ minutes. (Given $$\log 2 = 0.3010$$, $$\log 3 = 0.4771$$)
Round off your answer to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 1 - Chemistry - Question 60
The compound formed by the reaction of ethanal with semicarbazide contains _______ number of nitrogen atoms.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




