Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The rate of first order reaction is $$0.04 \text{ mol L}^{-1} \text{s}^{-1}$$ at $$10$$ minutes and $$0.03 \text{ mol L}^{-1} \text{s}^{-1}$$ at $$20$$ minutes after initiation. Half life of the reaction is ______ minutes. (Given $$\log 2 = 0.3010$$, $$\log 3 = 0.4771$$)
Round off your answer to the nearest integer.
Correct Answer: 24
For a first order reaction, the rate is given by:
$$ r = k[A] $$
Since the rate is proportional to concentration, the ratio of rates at two times gives the ratio of concentrations:
$$ \frac{r_1}{r_2} = \frac{[A]_1}{[A]_2} $$
Given: $$r_1 = 0.04$$ at $$t_1 = 10$$ min, $$r_2 = 0.03$$ at $$t_2 = 20$$ min.
$$ \frac{[A]_{10}}{[A]_{20}} = \frac{0.04}{0.03} = \frac{4}{3} $$
For a first order reaction, the rate constant is:
$$ k = \frac{2.303}{t_2 - t_1} \log\frac{[A]_1}{[A]_2} = \frac{2.303}{10} \log\frac{4}{3} $$
$$ k = \frac{2.303}{10}(\log 4 - \log 3) = \frac{2.303}{10}(2\log 2 - \log 3) $$
$$ k = \frac{2.303}{10}(2 \times 0.3010 - 0.4771) = \frac{2.303}{10}(0.6020 - 0.4771) $$
$$ k = \frac{2.303 \times 0.1249}{10} = \frac{0.2877}{10} = 0.02877 \text{ min}^{-1} $$
The half-life for a first order reaction is:
$$ t_{1/2} = \frac{0.693}{k} = \frac{0.693}{0.02877} \approx 24.09 \text{ min} $$
Rounding to the nearest integer: $$t_{1/2} \approx 24$$ minutes.
Therefore, the answer is $$\boxed{24}$$.
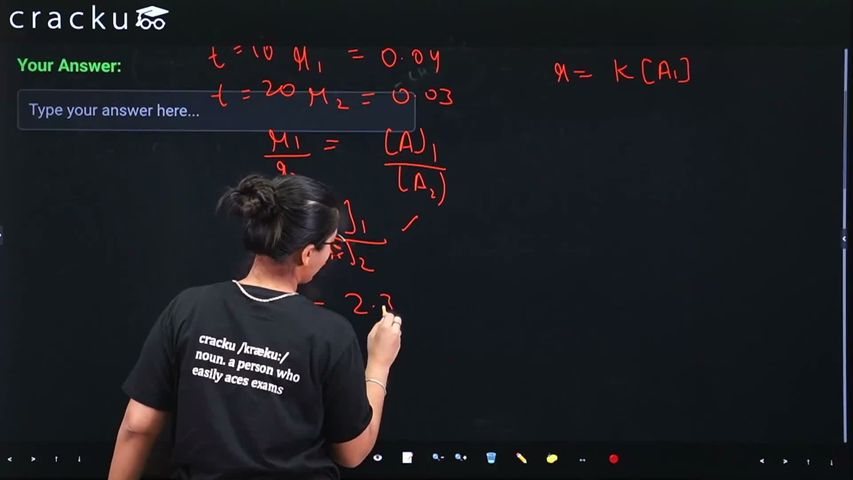
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
