Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The compound formed by the reaction of ethanal with semicarbazide contains _______ number of nitrogen atoms.
Correct Answer: 3
Ethanal ($$CH_3CHO$$) reacts with semicarbazide ($$NH_2NHCONH_2$$) to form a semicarbazone through a condensation reaction.
The reaction is:
$$ CH_3CHO + NH_2NHCONH_2 \rightarrow CH_3CH=NNHCONH_2 + H_2O $$
The product is acetaldehyde semicarbazone: $$CH_3CH=N-NH-CO-NH_2$$.
Counting the nitrogen atoms in the product:
1. The $$=N-$$ nitrogen
2. The $$-NH-$$ nitrogen
3. The $$-NH_2$$ nitrogen
Total nitrogen atoms = $$3$$.
Therefore, the answer is $$\boxed{3}$$.
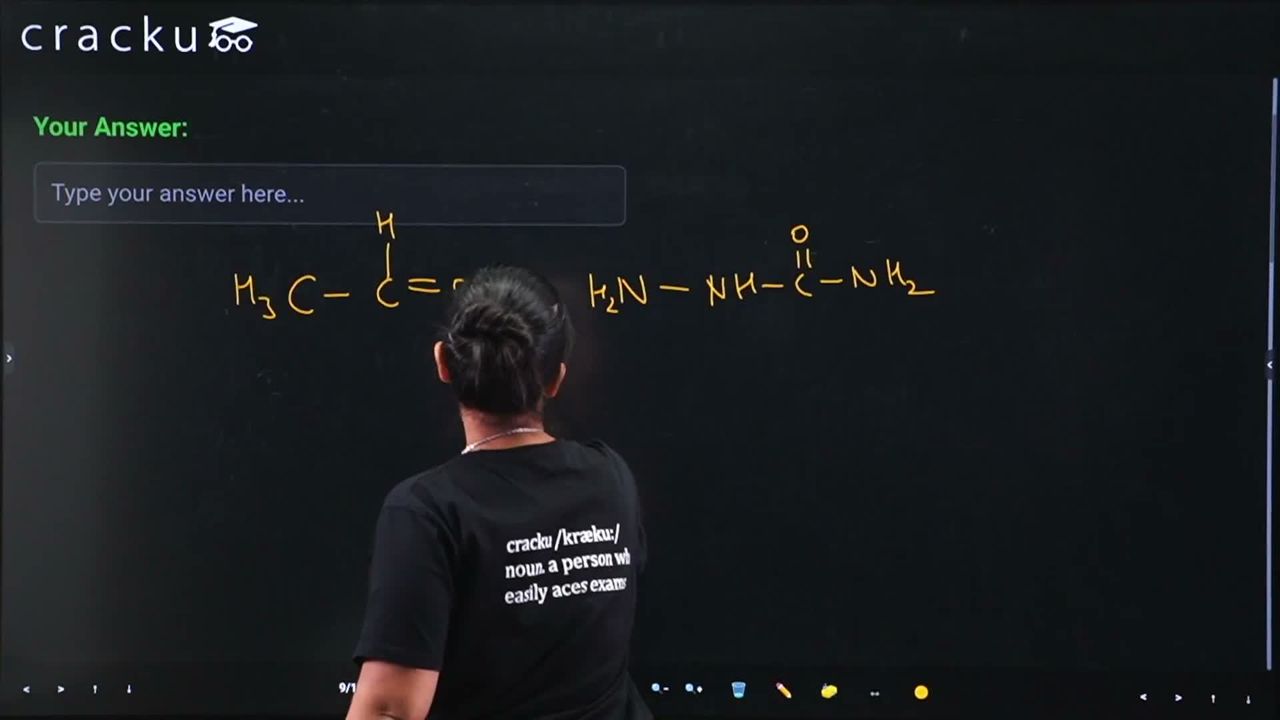
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
