NTA JEE Main 9th January 2019 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 31
For the following reaction, the mass of water produced from 445 g of $$C_{57}H_{110}O_6$$ is:
$$2 \; C_{57}H_{110}O_6(s) + 163O_2(g) \rightarrow 114 \; CO_2(g) + 110H_2O(l)$$
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 32
Which of the following combination of statements is true regarding the interpretation of the atomic orbitals?
(A) An electron in an orbital of high angular momentum stays away from the nucleus than an electron in the orbital of lower angular momentum.
(B) For a given value of the principal quantum number, the size of the orbit is inversely proportional to the azimuthal quantum number.
(C) According to wave mechanics, the ground state angular momentum is equal to $$\frac{h}{2\pi}$$.
(D) The plot of $$\psi$$ Vs $$r$$ for various azimuthal quantum numbers, shows peak shifting towards higher $$r$$ value.
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 33
When the first electron gain enthalpy $$(\Delta H_{eg})$$ of oxygen is $$-141$$ kJ/mol, its second electron gain enthalpy is:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 34
In which of the following processes, the bond order has increased and paramagnetic character has changed to diamagnetic?
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 35
The entropy change associated with the conversion of 1 kg of ice at 273 K to water vapours at 383 K is: (Specific heat of water liquid and water vapour are 4.2 kJ K$$^{-1}$$ and 2.0 kJ K$$^{-1}$$ kg$$^{-1}$$; heat of liquid fusion and vaporization of water are 334 kJ kg$$^{-1}$$ and 2491 kJ kg$$^{-1}$$, respectively). (log 273 = 2.436, log 373 = 2.572, log 383 = 2.583)
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 36
The temporary hardness of water is due to:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 37
The metal that forms nitride by reacting directly with $$N_2$$ of air is:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 38
Which of the following compounds is not aromatic?
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 39
The pH of rain water is approximately:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 40
Which of the following conditions in drinking water causes methemoglobinemia?
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 41
At 100$$^{\circ}$$C, copper (Cu) has FCC unit cell structure with cell edge length of $$x$$ $$\text{Å}$$. What is the approximate density of Cu (in g cm$$^{-3}$$) at this temperature? [Atomic Mass of Cu = 63.55 u]
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 42
A solution containing 62 g ethylene glycol in 250 g water is cooled to $$-10^{\circ}$$C. If $$K_f$$ for water is 1.86 K kg mol$$^{-1}$$, the amount of water (in g) separated as ice is:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 43
If the standard electrode potential for a cell is 2 V at 300 K, the equilibrium constant (K) for the reaction.
$$Zn(s) + Cu^{2+}(aq) \rightleftharpoons Zn^{2+}(aq) + Cu(s)$$
at 300 K is approximately: ($$R = 8$$ J K$$^{-1}$$ mol$$^{-1}$$, $$F = 96000$$ C mol$$^{-1}$$)
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 44
For the reaction, $$2A + B \rightarrow$$ products, when the concentration of $$A$$ and $$B$$ both were doubled, the rate of the reaction increased from 0.3 mol L$$^{-1}$$ s$$^{-1}$$ to 2.4 mol L$$^{-1}$$ s$$^{-1}$$. When the concentration of $$A$$ alone is doubled, the rate increased from 0.3 mol L$$^{-1}$$ s$$^{-1}$$ to 0.6 mol L$$^{-1}$$ s$$^{-1}$$. Which one of the following statements is correct?
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 45
Consider the following reversible chemical reactions:
$$A_2(g) + B_2(g) \stackrel{k_1}{\rightleftharpoons} 2AB(g) \ldots (1)$$
$$6AB(g) \stackrel{k_2}{\rightleftharpoons} 3A_2(g) + 3B_2(g) \ldots (2)$$
The relation between $$K_1$$ and $$K_2$$ is:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 46
For coagulation of arsenious sulphide sol, which of the following salt solutions will be most effective?
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 47
The correct match between Item I and Item II is:
Item I Item II
(A) Benzaldehyde (P) Mobile phase
(B) Alumina (Q) Adsorbent
(C) Acetonitrile (R) Adsorbate
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 48
The correct statement regarding the given Ellingham diagram is:
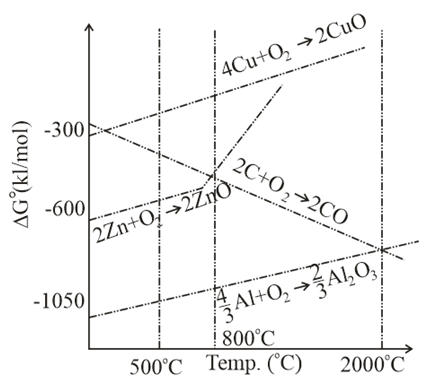
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 49
Good reducing nature of $$H_3PO_2$$ is attributed to the presence of:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 50
The transition element that has the lowest enthalpy of atomisation is:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 51
Homoleptic octahedral complexes of a metal ion $$M^{3+}$$ with three monodentate ligands $$L_1$$, $$L_2$$ and $$L_3$$ absorb wavelengths in the region of green, blue and red respectively. The increasing order of the ligand strength is:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 52
The complex that has highest crystal field splitting energy $$(\Delta)$$, is:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 53
The major product of the following reaction is:
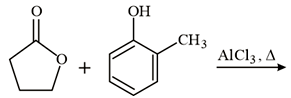
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 54
The products formed in the reaction of cumene with $$O_2$$ followed by treatment with dil HCl are:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 55
The major product formed in the following reaction is:
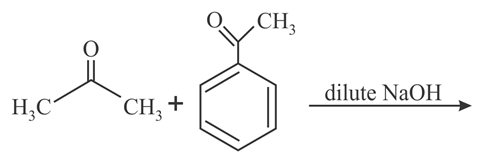
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 56
The test performed on compound x and their inferences are:
Test Inference
(a) 2,4-DNP test Coloured precipitate yellow
(b) Iodoform test Yellow precipitate
(c) Azo-dye test No dye formation
Compound 'x' is:
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 57
The major product obtained in the following reaction is:
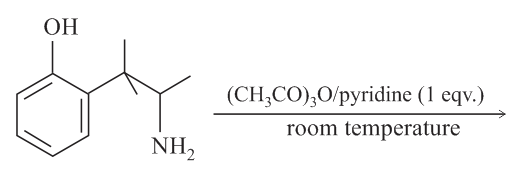
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 58
The increasing basicity order of the following compounds is:
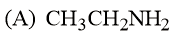
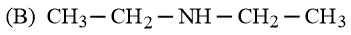
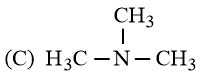
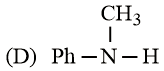
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 59
The major product of the following reaction is:
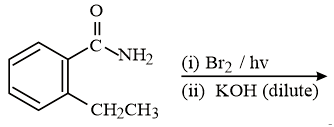
NTA JEE Main 9th January 2019 Shift 2 - Chemistry - Question 60
The correct sequence of amino acids present in the tripeptide given below is:
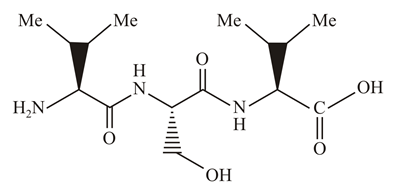
.webp)
.webp)
.webp)
.webp)




