NTA JEE Main 29th July 2022 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 31
$$N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)$$
Consider the reaction: . If 20 g of dinitrogen reacts with 5 g of dihydrogen, then the limiting reagent of the reaction and number of moles of $$NH_3$$ formed respectively are
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 32
The first ionization enthalpy of Na and Mg, respectively, are: 496 & 737 kJ mol$$^{-1}$$. The first ionization enthalpy (kJ mol$$^{-1}$$) of Al is
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 33
Which of the following pair of molecules contain odd electron molecule and an expanded octet molecule?
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 34
Number of lone pairs of electrons in the central atom of $$SCl_2$$, $$O_3$$, $$ClF_3$$ and $$SF_6$$, respectively, are
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 35
The reaction of zinc with excess of aqueous alkali, evolves hydrogen gas and gives
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 36
Lithium nitrate and sodium nitrate, when heated separately, respectively, give
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 37
Which among the following pairs of structures will give different products on ozonolysis? (Consider the double bonds in the structures are rigid and not delocalized.)
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 38
Which among the following pairs has only herbicides?
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 39
100 mL of 5% (w/v) solution of NaCl in water was prepared in 250 mL beaker. Albumin from the egg was poured into NaCl solution and stirred well. This resulted in a/an
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 40
In metallurgy the term "gangue" is used for
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 41
In following pairs, the one in which both transition metal ions are colourless is
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 42
In neutral or faintly alkaline medium, $$KMnO_4$$ being a powerful oxidant can oxidise, thiosulphate almost quantitatively, to sulphate. In this reaction overall change in oxidation state of manganese will be
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 43
Considering the above reactions, the compound 'A' and compound 'B' respectively are
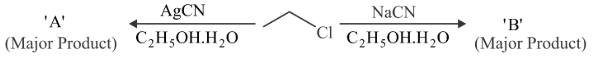
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 44
Consider the above reaction sequence, the Product 'C' is
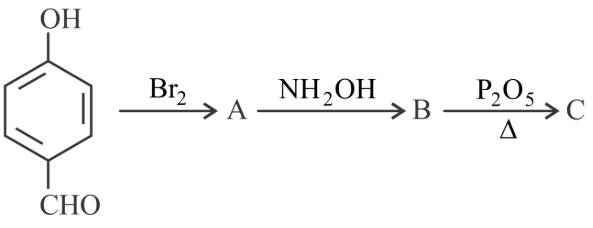
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 45
A compound 'X' is acidic and it is soluble in NaOH solution, but insoluble in $$NaHCO_3$$ solution. Compound 'X' also gives violet colour with neutral $$FeCl_3$$ solution. The compound 'X' is
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 46
Consider the following reaction sequence. The product 'B' is
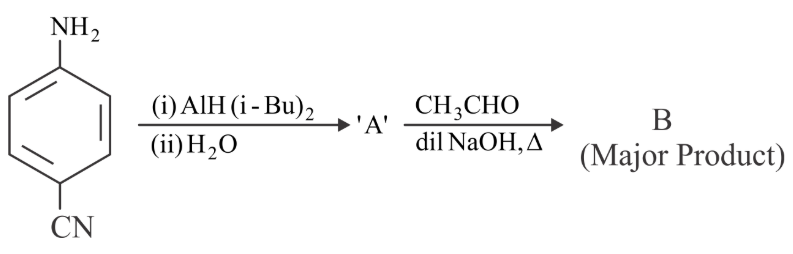
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 47
Which among the following is the strongest Bronsted base?
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 48
Consider the above reaction, the compound 'A' is
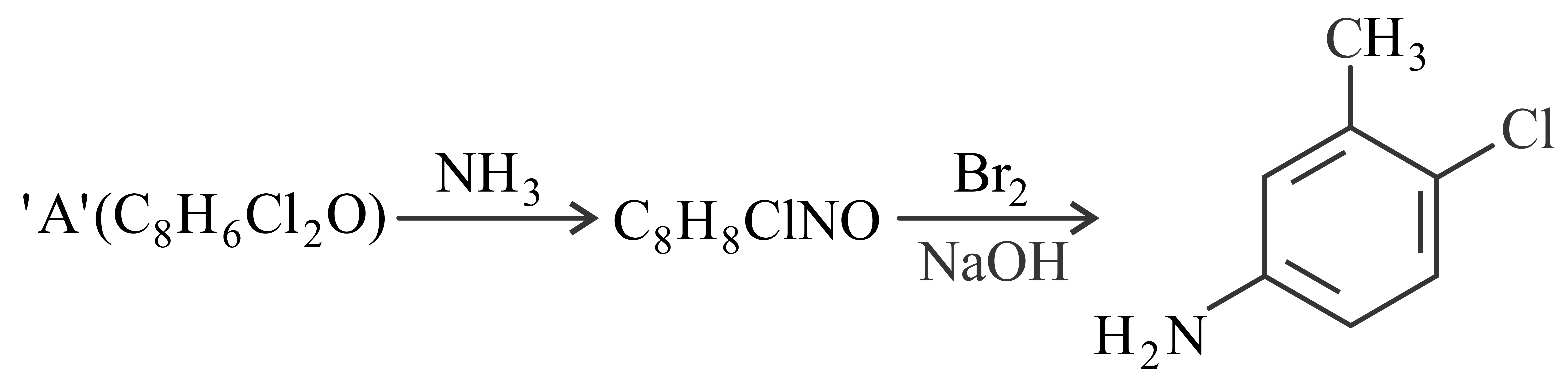
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 49
Which among the following represent reagent 'A'?
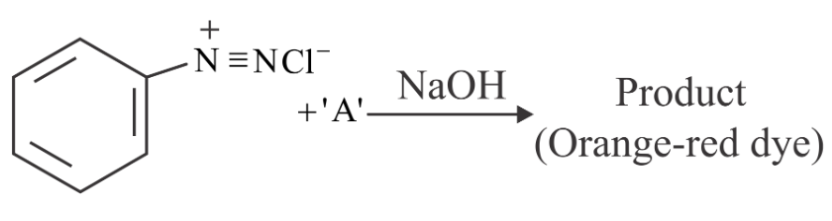
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 50
Which of the following compounds is an example of hypnotic drug?
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 51
The minimum uncertainty in the speed of an electron in one dimensional region of length $$2a_0$$ (Where $$a_0$$ = Bohr radius = 52.9 pm) is _____ km s$$^{-1}$$ (Nearest integer) (Given: Mass of electron $$= 9.1 \times 10^{-31}$$ kg, Planck's constant $$h = 6.63 \times 10^{-34}$$ Js)
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 52
When 600 mL of 0.2 M $$HNO_3$$ is mixed with 400 mL of 0.1 M NaOH solution in a flask, the rise in temperature of the flask is _____ $$\times 10^{-2}$$ °C (Enthalpy of neutralisation = 57 kJ mol$$^{-1}$$ and Specific heat of water = 4.2 J K$$^{-1}$$ g$$^{-1}$$) (Neglect heat capacity of flask)
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 53
If the solubility product of PbS is $$8 \times 10^{-28}$$, then the solubility of PbS in pure water at 298 K is $$x \times 10^{-16}$$ mol L$$^{-1}$$. The value of x is _____ (Nearest integer) [Given $$\sqrt{2} = 1.41$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 54
In bromination of Propyne, with Bromine 1,1,2,2-tetrabromopropane is obtained in 27% yield. The amount of 1,1,2,2-tetrabromopropane obtained from 1 g of Bromine in this reaction is _____ $$\times 10^{-1}$$ g. (Molar Mass: Bromine = 80 g/mol)
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 55
Ionic radii of cation $$A^+$$ and anion $$B^-$$ are 102 and 181 pm respectively. These ions are allowed to crystallize into an ionic solid. This crystal has cubic close packing for $$B^-$$. $$A^+$$ is present in all octahedral voids. The edge length of the unit cell of the crystal AB is _____ pm.
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 56
If $$O_2$$ gas is bubbled through water at 303 K, the number of millimoles of $$O_2$$ gas that dissolve in 1 litre of water is _____ (Nearest integer) (Given: Henry's Law constant for $$O_2$$ at 303 K is 46.82k bar and partial pressure of $$O_2$$ = 0.920 bar) (Assume solubility of $$O_2$$ in water is too small, nearly negligible)
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 57
Resistance of a conductivity cell (cell constant 129 m$$^{-1}$$) filled with 74.5 ppm solution of KCl is 100 $$\Omega$$ (labelled as solution 1). When the same cell is filled with KCl 149 ppm solution of KCl, the resistance is 50 $$\Omega$$ (labelled as solution 2). The ratio of molar conductivity of solution 1 and solution 2 is i.e. $$\frac{\Lambda_1}{\Lambda_2} = x \times 10^{-3}$$. The value of x is _____ (Given, molar mass of KCl is 74.5 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 58
The reaction between X and Y is first order with respect to X and zero order with respect to Y.
| Experiment | [X] mol L$$^{-1}$$ | [Y] mol L$$^{-1}$$ | Initial rate mol L$$^{-1}$$ min$$^{-1}$$ |
|---|---|---|---|
| I | 0.1 | 0.1 | $$2 \times 10^{-3}$$ |
| II | L | 0.2 | $$4 \times 10^{-3}$$ |
| III | 0.4 | 0.4 | $$M \times 10^{-3}$$ |
| IV | 0.1 | 0.2 | $$2 \times 10^{-3}$$ |
Examine the data of table and calculate ratio of numerical values of M and L.
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 59
$$[Fe(CN)_6]^{3-}$$ should be an inner orbital complex. Ignoring the pairing energy, the value of crystal field stabilization energy for this complex is $$(-) \Delta_0$$ _____ .
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 1 - Chemistry - Question 60
In a linear tetrapeptide (Constituted with different amino acids), (number of amino acids) - (number of peptide bonds) is
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




