Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
If $$O_2$$ gas is bubbled through water at 303 K, the number of millimoles of $$O_2$$ gas that dissolve in 1 litre of water is _____ (Nearest integer) (Given: Henry's Law constant for $$O_2$$ at 303 K is 46.82k bar and partial pressure of $$O_2$$ = 0.920 bar) (Assume solubility of $$O_2$$ in water is too small, nearly negligible)
Correct Answer: 1
We need to find the number of millimoles of $$O_2$$ that dissolve in 1 litre of water at 303 K using Henry's Law.
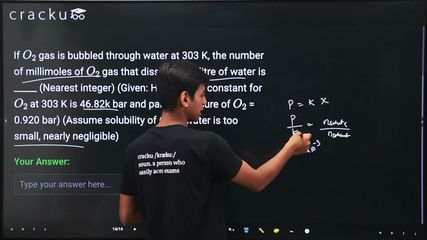
Henry's Law states: $$p = K_H \cdot x$$, where $$p$$ is the partial pressure of the gas, $$K_H$$ is the Henry's Law constant, and $$x$$ is the mole fraction of the gas in the solution.
Given: $$K_H = 46.82$$ kbar $$= 46.82 \times 10^3$$ bar, and $$p_{O_2} = 0.920$$ bar.
The mole fraction of $$O_2$$ in water is:
$$x = \frac{p}{K_H} = \frac{0.920}{46.82 \times 10^3} = \frac{0.920}{46820} = 1.9650 \times 10^{-5}$$
Now, since the solubility is very small, the mole fraction can be approximated as:
$$x \approx \frac{n_{O_2}}{n_{water}}$$
For 1 litre of water, $$n_{water} = \frac{1000}{18} = 55.556$$ mol.
$$n_{O_2} = x \times n_{water} = 1.9650 \times 10^{-5} \times 55.556 = 1.092 \times 10^{-3} \text{ mol}$$
Converting to millimoles: $$n_{O_2} = 1.092$$ mmol $$\approx 1$$ mmol (nearest integer).
Hence, the correct answer is 1.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
