NTA JEE Main 26th July 2022 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 31
Given two statements below:
Statement I: In $$Cl_2$$ molecule the covalent radius is double of the atomic radius of chlorine.
Statement II: Radius of anionic species is always greater than their parent atomic radius.
Choose the most appropriate answer from options given below
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 32
Match List - I with List - II.

Choose the correct answer from the options given below
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 33
Which of the given reactions is not an example of disproportionation reaction?
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 34
Which of the following can be used to prevent the decomposition of $$H_2O_2$$?
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 35
Reaction of $$BeCl_2$$ with $$LiAlH_4$$ gives:
(A) $$AlCl_3$$
(B) $$BeH_2$$
(C) $$LiH$$
(D) $$LiCl$$
(E) $$BeAlH_4$$
Choose the correct answer from options given below
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 36
Borazine, also known as inorganic benzene, can be prepared by the reaction of 3-equivalents of "X" with 6-equivalents of "Y". "X" and "Y", respectively are
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 37
Which technique among the following, is most appropriate in separation of a mixture of $$100 \text{ mg}$$ of $$p$$-nitrophenol and picric acid?
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 38
Which of the following compounds is not aromatic?
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 39
The correct stability order of the following diazonium salts is
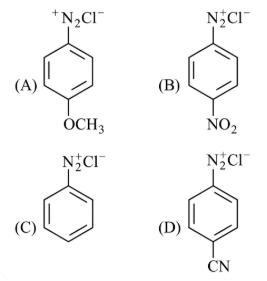
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 40
$$\dot{C}l + CH_4 \rightarrow A + B$$. $$A$$ and $$B$$ in the above atmospheric reaction step are
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 41
The major product formed in the given reaction is:
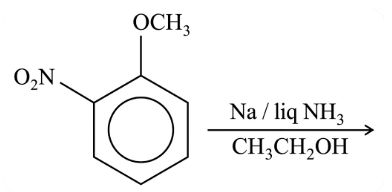
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 42
Match List - I with List - II.

Choose the correct answer from the options given below
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 43
Refining using liquation method is the most suitable for metals with
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 44
The dark purple colour of $$KMnO_4$$ disappears in the titration with oxalic acid in acidic medium. The overall change in the oxidation number of manganese in the reaction is
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 45
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): Experimental reaction of $$CH_3Cl$$ with aniline and anhydrous $$AlCl_3$$ does not give $$o$$ and $$p$$-methylaniline.
Reason (R): The $$-NH_2$$ group of aniline becomes deactivating because of salt formation with anhydrous $$AlCl_3$$ and hence yields $$m$$-methyl aniline as the product.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 46
The difference in the reaction of phenol with bromine in chloroform and bromine in water medium is due to
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 47
The products formed in the following reaction, A and B are
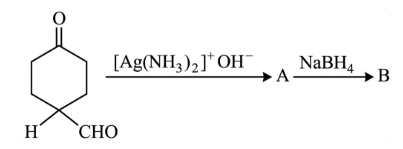
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 48
Which reactant will give the following alcohol on reaction with one mole of phenyl magnesium bromide (PhMgBr) followed by acidic hydrolysis?
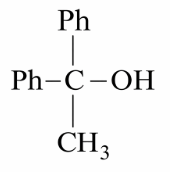
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 49
Stearic acid and polyethylene glycol react to form which one of the following soap/detergents?
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 50
Which of the following is a reducing sugar?
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 51
Chlorophyll extracted from the crushed green leaves was dissolved in water to make $$2 \text{ L}$$ solution of Mg of concentration $$48 \text{ ppm}$$. The number of atoms of Mg in this solution is $$x \times 10^{20}$$ atoms. The value of $$x$$ is ______ (Nearest Integer) (Given: Atomic mass of Mg is $$24 \text{ g mol}^{-1}$$, $$N_A = 6.02 \times 10^{23} \text{ mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 52
When $$800 \text{ mL}$$ of $$0.5M$$ nitric acid is heated in a beaker, its volume is reduced to half and $$11.5 \text{ g}$$ of nitric acid is evaporated. The molarity of the remaining nitric acid solution is $$x \times 10^{-2} \text{ M}$$. (Molar mass of nitric acid is $$63 \text{ g mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 53
The wavelength of an electron and a neutron will become equal when the velocity of the electron is $$x$$ times the velocity of neutron. The value of $$x$$ is ______ (the nearest integer) (Mass of electron is $$9.1 \times 10^{-31} \text{ kg}$$ and mass of neutron is $$1.6 \times 10^{-27} \text{ kg}$$)
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 54
A mixture of hydrogen and oxygen contains $$40\%$$ hydrogen by mass when the pressure is $$2.2 \text{ bar}$$. The partial pressure of hydrogen is ______ bar.
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 55
$$2.4 \text{ g}$$ coal is burnt in a bomb calorimeter in excess of oxygen at $$298 \text{ K}$$ and $$1 \text{ atm}$$ pressure. The temperature of the calorimeter rises from $$298 \text{ K}$$ to $$300 \text{ K}$$. The enthalpy change during the combustion of coal is $$-x \text{ kJ mol}^{-1}$$. The value of $$x$$ is ______ (Given: Heat capacity of bomb calorimeter $$20.0 \text{ kJ K}^{-1}$$. Assume coal to be pure carbon)
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 56
At $$298 \text{ K}$$, the equilibrium constant is $$2 \times 10^{15}$$ for the reaction:
$$Cu(s) + 2Ag^+(aq) \rightleftharpoons Cu^{2+}(aq) + 2Ag(s)$$
The equilibrium constant for the reaction
$$\dfrac{1}{2}Cu^{2+}(aq) + Ag(s) \rightleftharpoons \dfrac{1}{2}Cu(s) + Ag^+(aq)$$
is $$x \times 10^{-8}$$. The value of $$x$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 57
In the presence of sunlight, benzene reacts with $$Cl_2$$ to give product X. The number of hydrogens in X is ______.
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 58
The amount of charge in F (Faraday) required to obtain one mole of iron from $$Fe_3O_4$$ is ______. (Round off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 59
For a reaction $$A \rightarrow 2B + C$$, the half lives are $$100 \text{ s}$$ and $$50 \text{ s}$$ when the concentration of reactant A is $$0.5$$ and $$1.0 \text{ mol L =}$$ respectively. The order of the reaction is ______.
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 1 - Chemistry - Question 60
The difference between spin only magnetic moment values of $$[Co(H_2O)_6]Cl_2$$ and $$[Cr(H_2O)_6]Cl_3$$ is ______.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




