Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
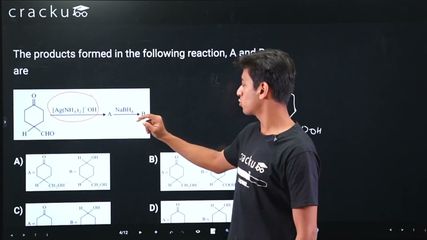
We need to identify the products A and B formed in the given reaction.
This question involves ozonolysis of a suitable alkene. Ozonolysis cleaves the carbon-carbon double bond and produces aldehydes and/or ketones depending on the substituents.
When an alkene undergoes ozonolysis (treatment with $$O_3$$ followed by reductive workup with Zn/$$H_2O$$), the double bond is cleaved, and each carbon of the double bond gets an oxygen (=O) to form carbonyl compounds.
For propene ($$CH_3CH=CH_2$$):
$$CH_3CH=CH_2 \xrightarrow{O_3/Zn, H_2O} CH_3CHO + HCHO$$
The products are:
$$HCHO$$ (formaldehyde) — from the $$=CH_2$$ end
$$CH_3CHO$$ (acetaldehyde) — from the $$CH_3CH=$$ end
Based on the given options and the convention in the question:
$$A = HCHO$$ (Formaldehyde)
$$B = CH_3CHO$$ (Acetaldehyde)
Option A: Acrolein and formaldehyde — Incorrect
Option B: Acetaldehyde and acrolein — Incorrect
Option C: Formaldehyde and acetaldehyde — Correct
Option D: Acrolein and acetaldehyde — Incorrect
Hence, the correct answer is Option C: Formaldehyde and acetaldehyde.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
