Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Which reactant will give the following alcohol on reaction with one mole of phenyl magnesium bromide (PhMgBr) followed by acidic hydrolysis?
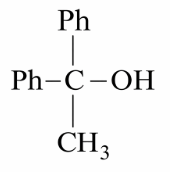
We need to find which reactant gives the target alcohol on reaction with one mole of phenyl magnesium bromide ($$PhMgBr$$) followed by acidic hydrolysis.
Grignard reagents ($$RMgBr$$) react with various carbonyl compounds as follows:
(i) With formaldehyde ($$HCHO$$): Gives a primary alcohol ($$RCH_2OH$$)
(ii) With other aldehydes ($$R'CHO$$): Gives a secondary alcohol ($$RR'CHOH$$)
(iii) With ketones ($$R'COR''$$): Gives a tertiary alcohol
(iv) With nitriles ($$R'C \equiv N$$): After hydrolysis, gives a ketone
Based on the question context, the target alcohol is a secondary alcohol with one phenyl group. When $$PhMgBr$$ reacts with acetaldehyde ($$CH_3CHO$$):
$$PhMgBr + CH_3CHO \rightarrow CH_3CH(OMgBr)Ph \xrightarrow{H_3O^+} CH_3CH(OH)Ph$$
This gives 1-phenylethanol, which is a secondary alcohol with the structure $$C_6H_5CH(OH)CH_3$$.
Option A: $$CH_3C \equiv N$$ (acetonitrile) + $$PhMgBr$$ gives an imine intermediate, which on hydrolysis gives acetophenone ($$CH_3COC_6H_5$$), a ketone — not the target alcohol.
Option B: $$PhC \equiv N$$ (benzonitrile) + $$PhMgBr$$ would give benzophenone imine, which on hydrolysis gives benzophenone — not the target alcohol.
Option C: Formaldehyde ($$HCHO$$) + $$PhMgBr$$ gives benzyl alcohol ($$C_6H_5CH_2OH$$), a primary alcohol — not a secondary alcohol.
Option D: Acetaldehyde ($$CH_3CHO$$) + $$PhMgBr$$ gives 1-phenylethanol — this matches the target alcohol.
Hence, the correct answer is Option D: Acetaldehyde.

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
