Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A disproportionation reaction is one in which the same element in a single oxidation state is simultaneously oxidized and reduced to form products with different oxidation states.
Option A: $$2H_2O_2 \rightarrow 2H_2O + O_2$$
Oxygen in H₂O₂ has oxidation state -1. In H₂O, oxygen is -2 (reduced). In O₂, oxygen is 0 (oxidized). This IS a disproportionation reaction.
Option B: $$2NO_2 + H_2O \rightarrow HNO_3 + HNO_2$$
Nitrogen in NO₂ has oxidation state +4. In HNO₃, nitrogen is +5 (oxidized). In HNO₂, nitrogen is +3 (reduced). This IS a disproportionation reaction.
Option C: $$MnO_4^- + 4H^+ + 3e^- \rightarrow MnO_2 + 2H_2O$$
Mn in MnO₄⁻ has oxidation state +7. In MnO₂, Mn is +4. This is a simple reduction reaction where Mn goes from +7 to +4 by gaining 3 electrons. There is no simultaneous oxidation and reduction of the same element. This is NOT a disproportionation reaction.
Option D: $$3MnO_4^{2-} + 4H^+ \rightarrow 2MnO_4^- + MnO_2 + 2H_2O$$
Mn in MnO₄²⁻ has oxidation state +6. In MnO₄⁻, Mn is +7 (oxidized). In MnO₂, Mn is +4 (reduced). This IS a disproportionation reaction.
The reaction that is NOT an example of a disproportionation reaction is Option C.
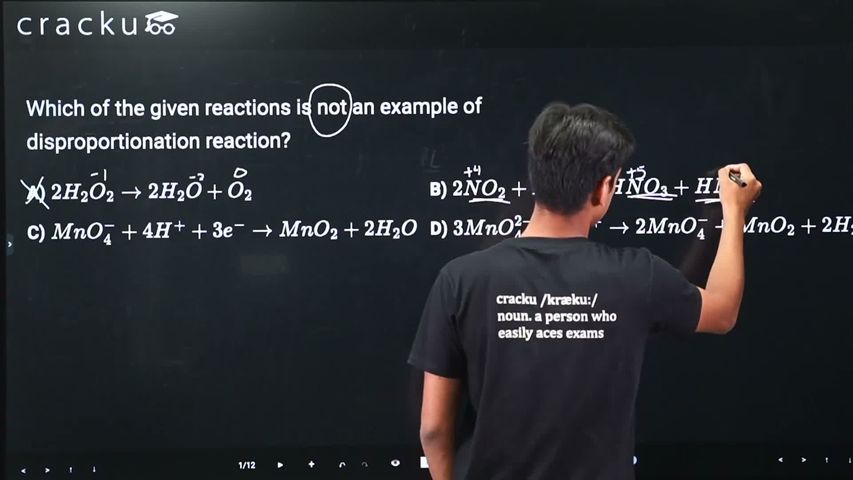
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
