NTA JEE Main 25th July 2022 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 31
$$SO_2Cl_2$$ on reaction with excess of water results into acidic mixture
$$SO_2Cl_2 + 2H_2O \rightarrow H_2SO_4 + 2HCl$$
16 moles of $$NaOH$$ is required for the complete neutralisation of the resultant acidic mixture. The number of moles of $$SO_2Cl_2$$ used is
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 32
Which of the following sets of quantum numbers is not allowed?
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 33
The IUPAC nomenclature of an element with electronic configuration $$[Rn] 5f^{14} 6d^1 7s^2$$ is
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 34
$$20 \text{ mL}$$ of $$0.1 \text{ M } NH_4OH$$ is mixed with $$40 \text{ mL}$$ of $$0.05 \text{ M HCl}$$. The pH of the mixture is nearest to:
(Given: $$K_b(NH_4OH) = 1 \times 10^{-5}$$, $$\log 2 = 0.30$$, $$\log 3 = 0.48$$, $$\log 5 = 0.69$$, $$\log 7 = 0.84$$, $$\log 11 = 1.04$$)
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 35
The reaction of $$H_2O_2$$ with potassium permanganate in acidic medium leads to the formation of mainly
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 36
Choose the correct order of density of the alkali metals
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 37
Match List - I with List - II
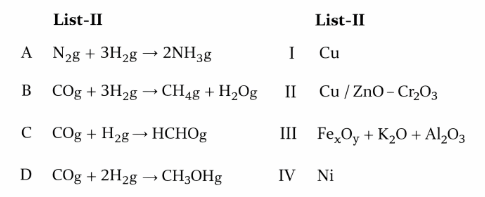
Choose the correct answer from the options given below
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 38
The geometry around boron in the product 'B' formed from the following reaction is
$$BF_3 + NaH \xrightarrow{450K} A + NaF$$
$$A + NMe_3 \rightarrow B$$
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 39
A compound 'A' on reaction with 'X' and 'Y' produces the same major product but different by-products 'a' and 'b'. Oxidation of 'a' gives a substance produced by ants.
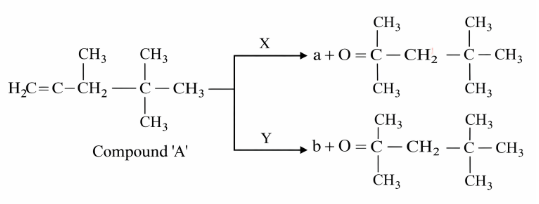
'X' and 'Y' respectively are
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 40
The photochemical smog does not generally contain
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 41
The depression in freezing point observed for a formic acid solution of concentration $$0.5 \text{ mL L}^{-1}$$ is $$0.0405°C$$. Density of formic acid is $$1.05 \text{ g mL}^{-1}$$. The Van't Hoff factor of the formic acid solution is nearly: (Given for water $$K_f = 1.86 \text{ K kg mol}^{-1}$$)
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 42
The compound(s) that is(are) removed as slag during the extraction of copper is:
(1) CaO
(2) FeO
(3) Al₂O₃
(4) ZnO
(5) NiO
Choose the correct answer from the options given below
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 43
The interhalogen compound formed from the reaction of bromine with excess of fluorine is a
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 44
Most stable product of the following reaction is
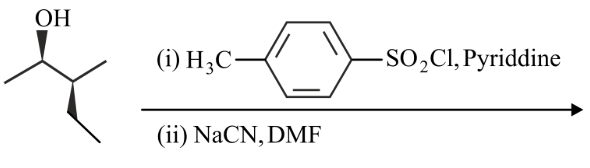
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 45
Given below are two statements:
Statement I: On heating with $$KHSO_4$$, glycerol is dehydrated and acrolein is formed.
Statement II: Acrolein has fruity odour and can be used to test glycerol's presence.
Choose the correct option.
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 46
Which one of the following reactions does not represent correct combination of substrate and product under the given conditions?
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 47
An organic compound 'A' on reaction with $$NH_3$$ followed by heating gives compound B. Which on further strong heating gives compound C ($$C_8H_5NO_2$$). Compound C on sequential reaction with ethanolic KOH, alkyl chloride and hydrolysis with alkali gives a primary amine. The compound A is
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 48
Melamine polymer is formed by the condensation of
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 49
Drugs used to bind to receptors, inhibiting its natural function and blocking a message are called
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 50
During the denaturation of proteins, which of these structures will remain intact?
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 51
Among the following species $$N_2, N_2^+, N_2^-, N_2^{2-}, O_2, O_2^+, O_2^-, O_2^{2-}$$, the number of species showing diamagnetism is ______.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 52
The pressure of a moist gas at $$27°C$$ is $$4 \text{ atm}$$. The volume of the container is doubled at the same temperature. The new pressure of the moist gas is ______ $$\times 10^{-1}$$ atm. (Nearest integer)
(Given: The vapour pressure of water at $$27°C$$ is $$0.4 \text{ atm}$$)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 53
The enthalpy of combustion of propane, graphite and dihydrogen at $$298 \text{ K}$$ are: $$-2220.0 \text{ kJ mol}^{-1}$$, $$-393.5 \text{ kJ mol}^{-1}$$ and $$-285.8 \text{ kJ mol}^{-1}$$ respectively. The magnitude of enthalpy of formation of propane ($$C_3H_8$$) is ______ $$\text{kJ mol}^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 54
While estimating the nitrogen present in an organic compound by Kjeldahl's method, the ammonia evolved from $$0.25 \text{ g}$$ of the compound neutralized $$2.5 \text{ mL}$$ of $$2M \text{ } H_2SO_4$$. The percentage of nitrogen present in organic compound is ______.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 55
The number of $$sp^3$$ hybridised carbons in an acyclic neutral compound with molecular formula $$C_4H_5N$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 56
The cell potential for $$Zn | Zn^{2+}(aq) || Sn^{x+} | Sn$$ is $$0.801 \text{ V}$$ at $$298 \text{ K}$$. The reaction quotient for the above reaction is $$10^{-2}$$. The number of electrons involved in the given electrochemical cell reaction is ______.
(Given $$E^0_{Zn^{2+}|Zn} = -0.763 \text{ V}$$, $$E^0_{Sn^{x+}|Sn} = +0.008 \text{ V}$$ and $$\dfrac{2.303RT}{F} = 0.06 \text{ V}$$)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 57
The half life for the decomposition of gaseous compound A is $$240 \text{ s}$$ when the gaseous pressure was $$500 \text{ Torr}$$ initially. When the pressure was $$250 \text{ Torr}$$, the half life was found to be $$4.0 \text{ min}$$. The order of the reaction is ______ (Nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 58
Among $$Co^{3+}$$, $$Ti^{2+}$$, $$V^{2+}$$ and $$Cr^{2+}$$ ions, one if used as a reagent cannot liberate $$H_2$$ from dilute mineral acid solution, its spin-only magnetic moment in gaseous state is ______ B.M. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 59
Consider the following metal complexes:
$$[Co(NH_3)]^{3+}$$
$$[CoCl(NH_3)_5]^{2+}$$
$$[Co(CN)_6]^{3-}$$
$$[Co(NH_3)_5(H_2O)]^{3+}$$
The spin-only magnetic moment value of the complex that absorbs light with shortest wavelength is ______ B.M. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 1 - Chemistry - Question 60
In the given reaction
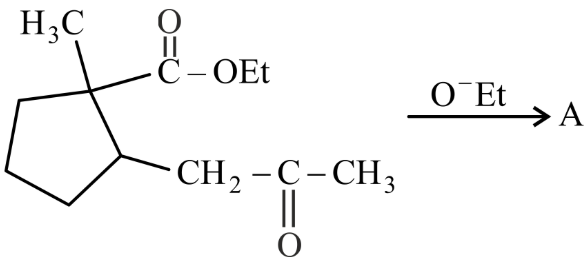
(Where Et is $$-C_2H_5$$)
The number of chiral carbon(s) in product A is ______.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




