NTA JEE Main 10th January 2019 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 31
Which of the graphs shown below does not represent the relationship between incident light and the electron ejected from metal surface?
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 32
The type of hybridization and no. of lone pair(s) of electron of Xe in $$XeOF_4$$, respectively, are:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 33
Two pi and half sigma bonds are present in:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 34
A process has $$\Delta H = 200$$ J mol$$^{-1}$$ and $$\Delta S = 40$$ J K$$^{-1}$$ mol$$^{-1}$$. Out of the values given below choose the minimum temperature above which the process will be spontaneous:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 35
What are the values of $$\frac{K_p}{K_c}$$ for the following reactions at 300 K respectively?
(At 300 K, RT = 24.62 dm$$^2$$ atm mol$$^{-1}$$)
$$N_2(g) + O_2(g) \rightleftharpoons 2NO(g)$$
$$N_2O_4(g) \rightleftharpoons 2NO(g)$$
$$N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)$$
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 36
A mixture of 100 m mol of $$Ca(OH)_2$$ and 2 g of sodium sulphate was dissolved in water and the volume was made up to 100 mL. What is the mass of calcium sulphate formed and the concentration of $$OH^-$$ in resulting solution, respectively? (Molar mass of $$Ca(OH)_2$$, $$Na_2SO_4$$ and $$CaSO_4$$ are 74, 143 and 136 g mol$$^{-1}$$, respectively; $$K_{sp}$$ of $$Ca(OH)_2$$ is $$5.5 \times 10^{-6}$$)
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 37
The chemical nature of hydrogen peroxide is:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 38
The total number of isotopes of hydrogen and number of radioactive isotopes among them, respectively, are:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 39
The metal used for making X-ray tube window is:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 40
The electronegativity of aluminum is similar to:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 41
The increasing order of the pKa values of the following compounds is:
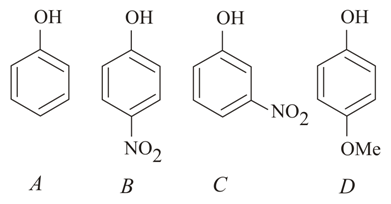
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 42
If Dichloromethane (DCM) and water $$H_2O$$ are used for differential extraction, which one of the following statements is correct?
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 43
The major product of the following reaction is:
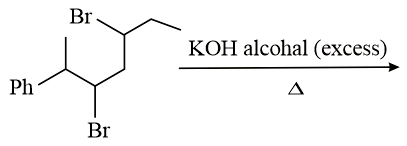
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 44
Which hydrogen in compound (E) is easily replaceable during bromination reaction in presence of light?
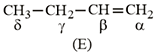
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 45
Water filled in two glasses A and B gave BOD values of 10 and 20, respectively. The correct statement regarding them is:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 46
Which primitive unit cell has unequal edge lengths ($$a \neq b \neq c$$) and all axial angles different from 90$$^{\circ}$$?
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 47
Liquids A and B form an ideal solution in the entire composition range. At 350K, the vapour pressure of pure A and pure B are $$7 \times 10^3$$ Pa and $$12 \times 10^3$$ Pa, respectively. The composition of the vapour in equilibrium with a solution containing 40 mole percent of A at this temperature is:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 48
Consider the following reduction processes:
$$Zn^{2+} + 2e^- \rightarrow Zn(s); E^{\circ} = -0.76$$ V
$$Ca^{2+} + 2e^- \rightarrow Ca(s); E^{\circ} = -2.87$$ V
$$Mg^{2+} + 2e^- \rightarrow Mg(s); E^{\circ} = -2.36$$ V
$$Ni^{2+} + 2e^- \rightarrow Ni(s); E^{\circ} = -0.25$$ V
The reducing power of the metals increases in the order:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 49
Consider the given plots for a reaction obeying Arrhenius equation ($$0^{\circ}$$C < T < 300$$^{\circ}$$C): (K and $$E_a$$ are rate constant and activation energy, respectively)
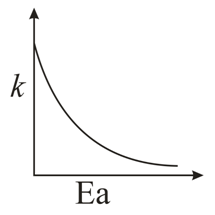
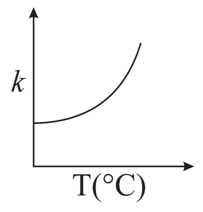
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 50
Which of the following is not an example of heterogeneous catalysis reaction?
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 51
Hall Heroult's process is given by:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 52
The effect of lanthanoid contraction in the lanthanoid series of elements by and large means:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 53
The total number of isomers for a square planar complex: $$[MCl(F)(NO_2)(SCN)]$$ is:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 54
Wilkinson catalyst is:
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 55
The major product of the following reaction is:
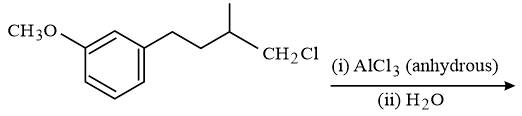
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 56
The major product 'X' formed in the following reaction is:
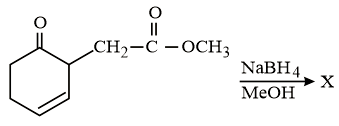
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 57
The decreasing order of ease of alkaline hydrolysis for the following esters is:
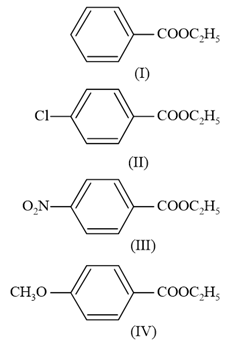
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 58
With dehydrating agent present which dicarboxylic acid is least reactive towards forming an anhydride?
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 59
The major product formed in the reaction given below will be:
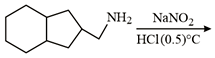
NTA JEE Main 10th January 2019 Shift 1 - Chemistry - Question 60
The correct structure of the product 'P' in the following reaction is
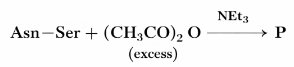
.webp)
.webp)
.webp)
.webp)




