Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
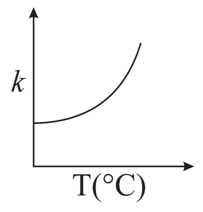
Consider the given plots for a reaction obeying Arrhenius equation ($$0^{\circ}$$C < T < 300$$^{\circ}$$C): (K and $$E_a$$ are rate constant and activation energy, respectively)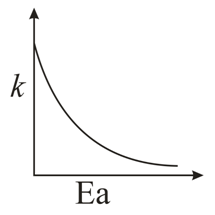
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.