NTA JEE Main 29th July 2022 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 31
Consider the reaction
$$4HNO_3(l) + 3KCl(s) \to Cl_2(g) + NOCl(g) + 2H_2O(g) + 3KNO_3(s)$$
The amount of $$HNO_3$$ required to produce 110.0 g of $$KNO_3$$ is (Given: Atomic masses of H, O, N and K are 1, 16, 14 and 39, respectively.)
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 32
$$C(s) + O_2(g) \to CO_2(g) + 400$$ kJ
$$C(s) + \frac{1}{2}O_2(g) \to CO(g) + 100$$ kJ
When coal of purity 60% is allowed to burn in presence of insufficient oxygen, 60% of carbon is converted into 'CO' and the remaining is converted into '$$CO_2$$'. The heat generated when 0.6 kg of coal is burnt is
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 33
Given below are the quantum numbers for 4 electrons.
A. $$n=3, l=2, m_l=1, m_s=+\frac{1}{2}$$
B. $$n=4, l=1, m_l=0, m_s=+\frac{1}{2}$$
C. $$n=4, l=2, m_l=-2, m_s=-\frac{1}{2}$$
D. $$n=3, l=1, m_l=-1, m_s=+\frac{1}{2}$$
The correct order of increasing energy is
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 34
200 mL of 0.01 M HCl is mixed with 400 mL of 0.01 M $$H_2SO_4$$. The pH of the mixture is
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 35
A compound 'X' is a weak acid and it exhibits colour change at pH close to the equivalence point during neutralization of NaOH with $$CH_3COOH$$. Compound 'X' exists in ionized form in basic medium. The compound 'X' is
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 36
Given below are two statements.
Statement I: Stannane is an example of a molecular hydride.
Statement II: Stannane is a planar molecule.
In the light of the above statement, choose the most appropriate answer from the options given below
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 37
Portland cement contains 'X' to enhance the setting time. What is 'X'?
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 38
When borax is heated with CoO on a platinum loop, blue coloured bead formed is largely due to
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 39
Correct structure of $$\gamma$$-methylcyclohexane carbaldehyde is
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 40
Given below are two statements.
Statement I: The compound is optically active.
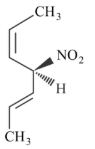
Statement II: The second compound is mirror image of the above compound A.
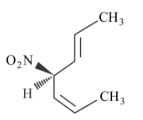
In the light of the above statement, choose the most appropriate answer from the options given below.
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 41
Given below are the critical temperatures of some of the gases:
| Gas | Critical temperature (K) |
|---|---|
| He | 5.2 |
| $$CH_4$$ | 190 |
| $$CO_2$$ | 304.2 |
| $$NH_3$$ | 405.5 |
The gas showing least adsorption on a definite amount of charcoal is
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 42
In liquation process used for tin (Sn), the metal
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 43
Dinitrogen is a robust compound, but reacts at high altitude to form oxides. The oxide of nitrogen that can damage plant leaves and retard photosynthesis is
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 44
Which of the following 3d-metal ion will give the lowest enthalpy of hydration $$\Delta_{hyd}H$$ when dissolved in water?
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 45
Octahedral complexes of copper(II) undergo structural distortion (Jahn-Teller). Which one of the given copper(II) complexes will show the maximum structural distortion? (en = ethylenediamine; $$H_2N-CH_2-CH_2-NH_2$$)
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 46
Compound 'A' undergoes following sequence of reactions to give compound 'B'. The correct structure and chirality of compound 'B' is [where Et is $$-C_2H_5$$]
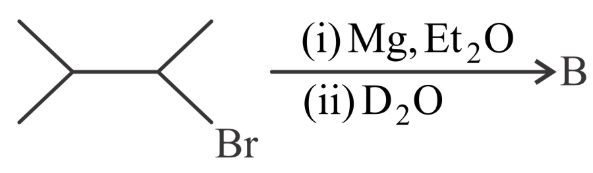
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 47
When ethanol is heated with conc. $$H_2SO_4$$, a gas is produced. The compound formed, when this gas is treated with cold dilute aqueous solution of Baeyer's reagent, is
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 48
The Hinsberg reagent is
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 49
Which of the following is NOT a natural polymer?
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 50
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Amylose is insoluble in water.
Reason R: Amylose is a long linear molecule with more than 200 glucose units.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 51
A 1.84 mg sample of polyhydric alcoholic compound 'X' of molar mass 92.0 g/mol gave 1.344 mL of $$H_2$$ gas at STP. The number of alcoholic hydrogen present in compound 'X' is
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 52
Consider, $$PF_5, BrF_5, PCl_3, SF_6, ICl_4^-, ClF_3$$ and $$IF_5$$. Amongst the above molecule(s)/ion(s), the number of molecule(s)/ion(s) having $$sp^3d^2$$ hybridisation is
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 53
'x' g of molecular oxygen $$(O_2)$$ is mixed with 200 g of neon (Ne). The total pressure of the non-reactive mixture of $$O_2$$ and Ne in the cylinder is 25 bar. The partial pressure of Ne is 20 bar at the same temperature and volume. The value of 'x' is [Given: Molar mass of $$O_2$$ = 32 g mol$$^{-1}$$. Molar mass of Ne = 20 g mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 54
1.80 g of solute A was dissolved in 62.5 cm$$^3$$ of ethanol and freezing point of the solution was found to be 155.1 K. The molar mass of solute A is _____ g mol$$^{-1}$$. [Given: Freezing point of ethanol is 156.0 K. Density of ethanol is 0.80 g cm$$^{-3}$$. Freezing point depression constant of ethanol is 2.00 K kg mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 55
For a cell, $$Cu(s)|Cu^{2+}(0.001 M)||Ag^+(0.01 M)|Ag(s)$$, the cell potential is found to be 0.43 V at 298 K. The magnitude of standard electrode potential for $$Cu^{2+}|Cu$$ is _____ $$\times 10^{-2}$$ V. Given: $$E^\theta_{Ag^+/Ag} = 0.80$$ V and $$\frac{2.303RT}{F} = 0.06$$ V
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 56
Assuming 1 $$\mu$$g of trace radioactive element X with a half life of 30 years is absorbed by a growing tree. The amount of X remaining in the tree after 100 years is _____ $$\times 10^{-1}$$ $$\mu$$g. [Given: $$\ln 10 = 2.303$$; $$\log 2 = 0.30$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 57
Consider the following sulphur based oxoacids. $$H_2SO_3, H_2SO_4, H_2S_2O_8$$ and $$H_2S_2O_7$$. Amongst these oxoacids, the number of those with peroxo (O-O) bond is
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 58
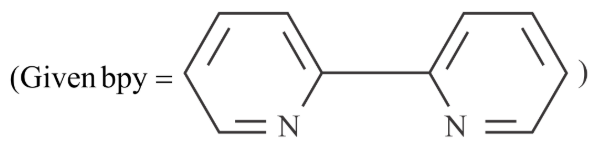
Sum of oxidation state (magnitude) and coordination number of cobalt in $$Na[Co(bpy)Cl_4]$$ is
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 59
The number of stereoisomers formed in a reaction of $$[\pm] PhC(=O)C(OH)(CN)Ph$$ with HCN is
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Chemistry - Question 60
The number of chlorine atoms in bithionol is
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




