NTA JEE Main 24th June 2022 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 31
$$120$$ g of an organic compound which contains only carbon and hydrogen on complete combustion gives $$330$$ g of $$CO_2$$ and $$270$$ g of water. The percentage of carbon and hydrogen in the organic compound are respectively
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 32
The energy of one mole of photons of radiation of wavelength $$300$$ nm is (Given :
$$h = 6.63 \times 10^{-34}$$ J s, $$N_A = 6.02 \times 10^{23}$$ mol$$^{-1}$$, $$c = 3 \times 10^{8}$$ m s$$^{-1}$$)
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 33
Metals generally melt at very high temperatures, Among the following which one has the highest melting point?
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 34
The correct order of bond orders of $$C_2^{2-}$$, $$N_2^{2-}$$ and $$O_2^{2-}$$ is
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 35
At $$25°$$C and 1 atm pressure, the enthalpies of combustion are as given below:
| Substance | $$H_2$$ | C (graphite) | $$C_2H_6(g)$$ |
|---|---|---|---|
| $$\frac{\Delta_c H^\ominus}{kJ mol^{-1}}$$ | $$-286.0$$ | $$-394.0$$ | $$-1560.0$$ |
The enthalpy of formation of ethane is
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 36
Which one of the following compounds is used as a chemical in certain type of fire extinguishers
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 37
Arrange the following carbocations in decreasing order of stability.
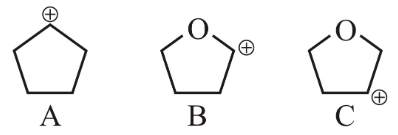
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 38
Given below are two statements.
Statement I: The presence of weaker $$\pi$$-bonds make alkenes less stable than alkanes
Statement II: The strength of the double bond is greater than that of carbon-carbon single bond.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 39
Which of the following reagents / reactions will convert 'A' to 'B'?
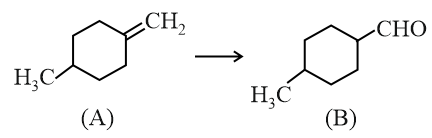
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 40
Some gases are responsible for heating of atmosphere (green house effect). Identify from the following the gaseous species which does not cause it.
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 41
In the industrial production of which of the following, molecular hydrogen is obtained as a bye product.
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 42
For a first order reaction, the time required for completion of $$90\%$$ reaction is '$$x$$' times the half life of the reaction. The value of '$$x$$' is
(Given: $$\ln 10 = 2.303$$ and $$\log 2 = 0.3010$$)
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 43
Which of the following chemical reactions represents Hall-Heroult Process?
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 44
$$PCl_5$$ is well known but $$NCl_5$$ is not. Because,
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 45
Transition metal complex with highest value of crystal field splitting $$(\Delta_0)$$ will be
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 46
Hex-4-ene-2-ol on treatment with PCC gives 'A'. 'A' on reaction with sodium hypoiodite gives 'B', which on further heating with soda lime gives 'C'. The compound 'C' is
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 47
The conversion of propan-1-ol to n-butylamine involves the sequential addition of reagents. The correct sequential order of reagents is
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 48
Which of the following is not a condensation polymer?
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 49
The structure shown below is of which well-known drug molecule?
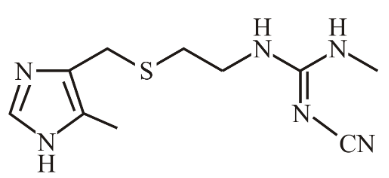
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 50
In the flame test of a mixture of salts, a green flame with blue centre was observed. Which one of the following cations may be present?
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 51
At $$300$$ K, a sample of $$3.0$$ g of gas A occupies the same volume as $$0.2$$ g of hydrogen at $$200$$ K at the same pressure. The molar mass of gas A is ______ g mol$$^{-1}$$. (nearest integer) Assume that the behaviour of gases as ideal.
(Given: The molar mass of hydrogen ($$H_2$$) gas is $$2.0$$ g mol$$^{-1}$$.)
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 52
$$PCl_5$$ dissociates as $$PCl_5(g) \rightleftharpoons PCl_3(g) + Cl_2(g)$$. $$5$$ moles of $$PCl_5$$ are placed in a $$200$$ litre vessel which contains $$2$$ moles of $$N_2$$ and is maintained at $$600$$ K. The equilibrium pressure is $$2.46$$ atm. The equilibrium constant $$K_p$$ for the dissociation of $$PCl_5$$ is ______ $$\times 10^{-3}$$. (nearest integer) (Given: $$R = 0.082$$ L atm K$$^{-1}$$ mol$$^{-1}$$; Assume ideal gas behaviour)
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 53
Manganese (VI) has ability to disproportionate in acidic solution. The difference in oxidation states of two ions it forms in acidic solution is ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 54
$$0.2$$ g of an organic compound was subjected to estimation of nitrogen by Dumas method in which volume of $$N_2$$ evolved (at STP) was found to be $$22.400$$ mL. The percentage of nitrogen in the compound is ______ [nearest integer] (Given: Molar mass of $$N_2$$ is $$28$$ g mol$$^{-1}$$, Molar volume of $$N_2$$ at STP: $$22.4$$ L)
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 55
A company dissolves '$$x$$' amount of $$CO_2$$ at $$298$$ K in $$1$$ litre of water to prepare soda water. $$X = $$ ______ $$\times 10^{-3}$$ g. (nearest integer)
(Given: partial pressure of $$CO_2$$ at $$298$$ K $$= 0.835$$ bar. Henry's law constant for $$CO_2$$ at $$298$$ K $$= 1.67$$ kbar. Atomic mass of H, C and O is $$1, 12,$$ and $$6$$ g mol$$^{-1}$$, respectively)
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 56
The resistance of a conductivity cell containing $$0.01$$ MKCl solution at $$298$$ K is $$1750$$ $$\Omega$$. If the conductivity of $$0.01$$ MKCl solution at $$298$$ K is $$0.152 \times 10^{-3}$$ S cm$$^{-1}$$, then the cell constant of the conductivity cell is ______ $$\times 10^{-3}$$ cm$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 57
When $$200$$ mL of $$0.2$$ M acetic acid is shaken with $$0.6$$ g of wood charcoal, the final concentration of acetic acid after adsorption is $$0.1$$ M. The mass of acetic acid adsorbed per gram of carbon is ______ g.
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 58
(a) Baryte, (b) Galena, (c) Zinc blende and (d) Copper pyrites. How many of these minerals are sulphide based? ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 59
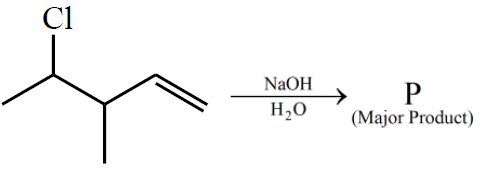
Consider the above reaction. The number of $$\pi$$ electrons present in the product 'P' is ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Chemistry - Question 60
In alanylglycylleucylalanylvaline the number of peptide linkages is: ______
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




