Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Manganese (VI) has ability to disproportionate in acidic solution. The difference in oxidation states of two ions it forms in acidic solution is ______
Correct Answer: 3
We need to find the difference in oxidation states of the two ions formed when Manganese(VI) disproportionates in acidic solution. Manganese in the +6 oxidation state exists as the manganate ion: $$MnO_4^{2-}$$ (where Mn is +6).
In disproportionation, the same species is simultaneously oxidized and reduced. In acidic solution, $$MnO_4^{2-}$$ disproportionates as:
$$3MnO_4^{2-} + 4H^+ \to 2MnO_4^- + MnO_2 + 2H_2O$$
In $$MnO_4^-$$ (permanganate ion): Let the oxidation state of Mn be $$y$$. Then $$y + 4(-2) = -1$$, giving $$y = +7$$. In $$MnO_2$$ (manganese dioxide): Let the oxidation state of Mn be $$z$$. Then $$z + 2(-2) = 0$$, giving $$z = +4$$.
Difference in oxidation states = $$+7 - (+4) = 3$$. The answer is 3.
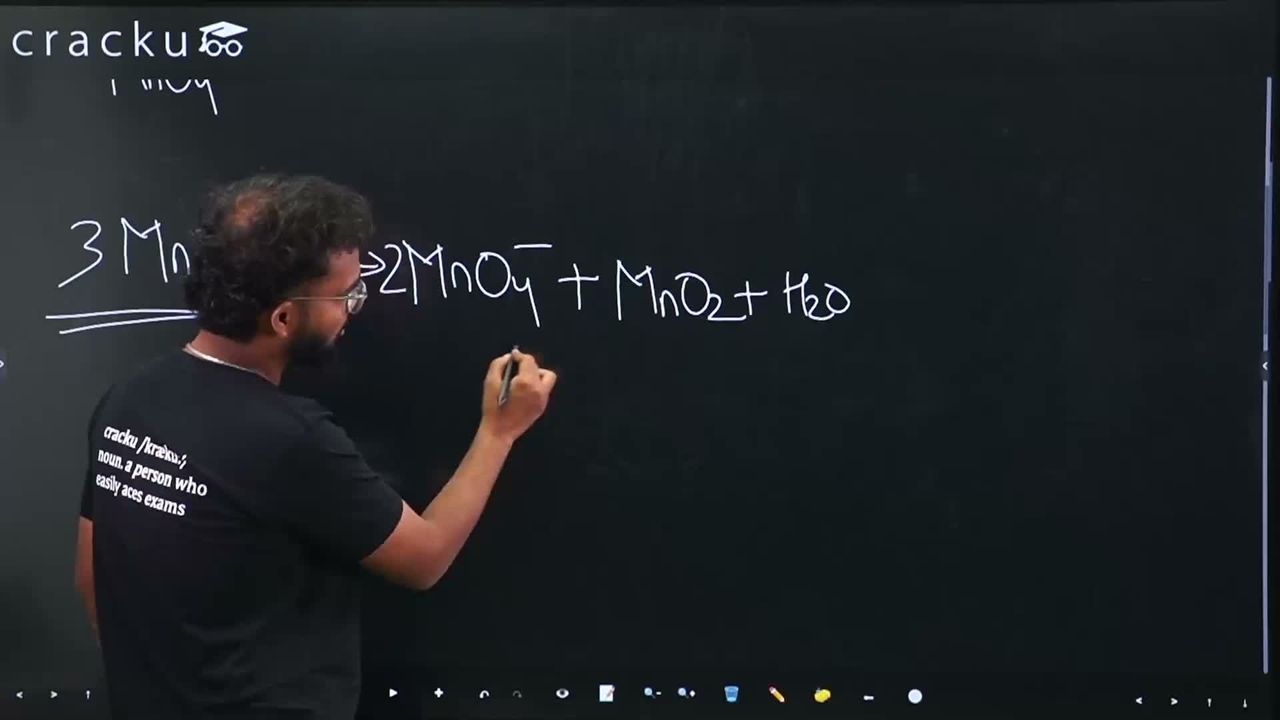
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
