Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
$$0.2$$ g of an organic compound was subjected to estimation of nitrogen by Dumas method in which volume of $$N_2$$ evolved (at STP) was found to be $$22.400$$ mL. The percentage of nitrogen in the compound is ______ [nearest integer] (Given: Molar mass of $$N_2$$ is $$28$$ g mol$$^{-1}$$, Molar volume of $$N_2$$ at STP: $$22.4$$ L)
Correct Answer: 14
We are given that 0.2 g of an organic compound produces $$N_2$$ gas with volume 22.400 mL at STP using the Dumas method.
Since the volume of $$N_2$$ must be expressed in litres, we have:
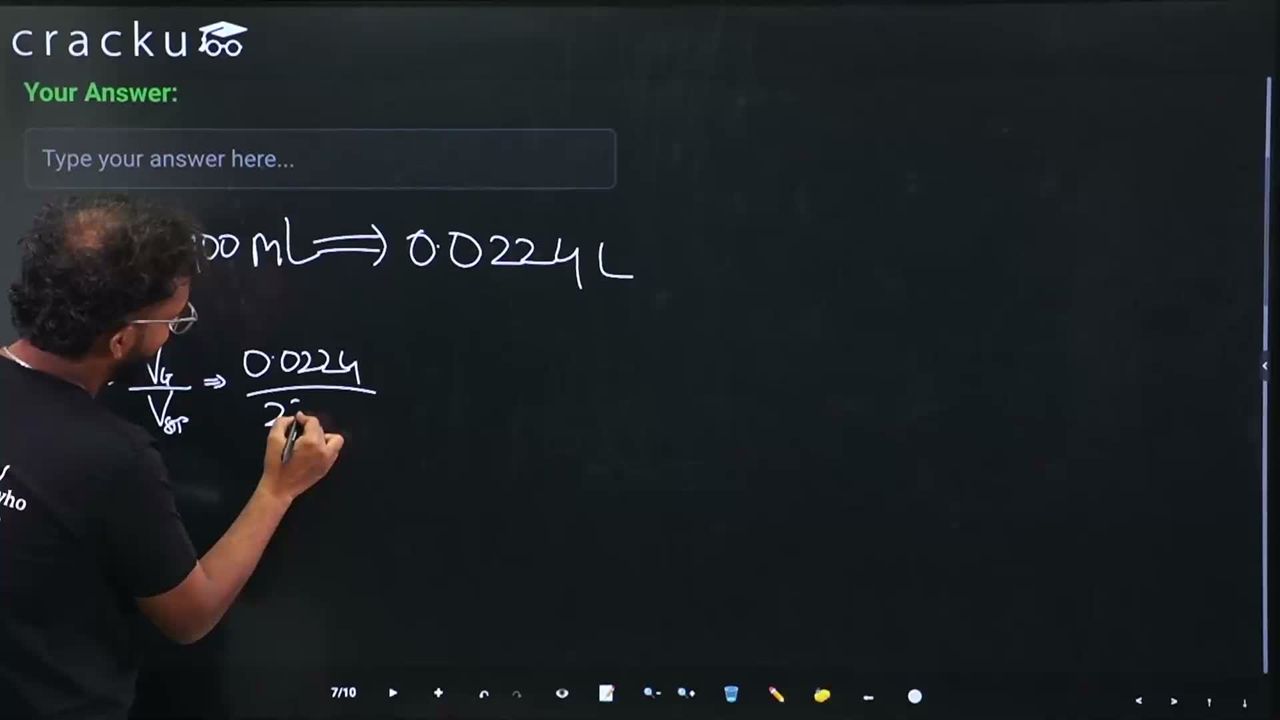
$$ V_{N_2} = 22.400 \text{ mL} = 0.02240 \text{ L} $$
Substituting this into the molar volume relation at STP (22.4 L) gives:
$$ n_{N_2} = \frac{V}{V_m} = \frac{0.02240}{22.4} = 0.001 \text{ mol} $$
This yields the mass of nitrogen as:
$$ m_{N_2} = n \times M = 0.001 \times 28 = 0.028 \text{ g} $$
From the above, the percentage of nitrogen in the compound is:
$$ \% N = \frac{m_{N_2}}{m_{\text{compound}}} \times 100 = \frac{0.028}{0.2} \times 100 = 14\% $$
Therefore, the percentage of nitrogen in the compound is 14.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
