Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
We need to explain why $$PCl_5$$ exists but $$NCl_5$$ does not.
To form five bonds (as in $$PCl_5$$), the central atom needs five orbitals for bonding, which requires $$sp^3d$$ hybridization. This hybridization uses one d-orbital.
Phosphorus (P): Electronic configuration is $$[Ne]\, 3s^2\, 3p^3$$. Phosphorus is in the third period and has access to vacant 3d orbitals. It can undergo $$sp^3d$$ hybridization to form 5 bonds, giving $$PCl_5$$ a trigonal bipyramidal structure.
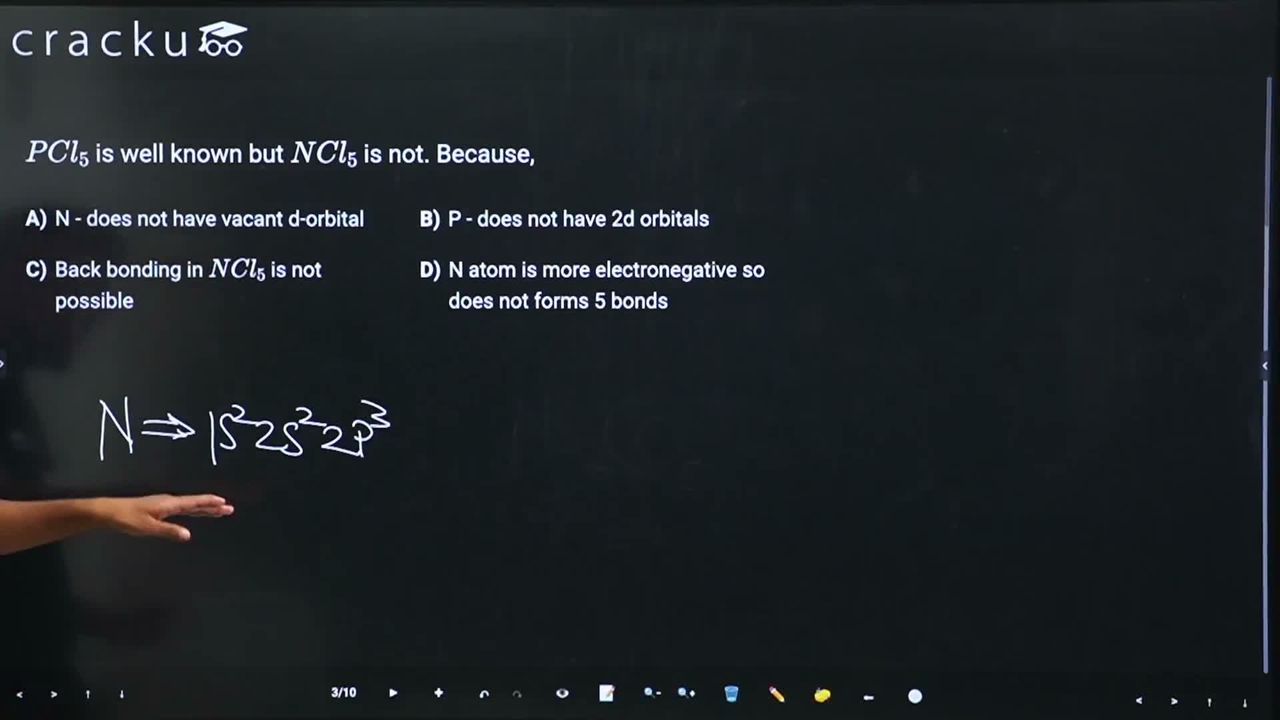
Nitrogen (N): Electronic configuration is $$[He]\, 2s^2\, 2p^3$$. Nitrogen is in the second period and has only 2s and 2p orbitals in its valence shell. There are no 2d orbitals available (d-orbitals start from the 3rd shell). Therefore, nitrogen cannot expand its octet and cannot form 5 bonds.
Since nitrogen lacks vacant d-orbitals, it can form a maximum of 4 bonds and cannot accommodate 5 chlorine atoms.
The correct answer is Option A: N does not have vacant d-orbital.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
