Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
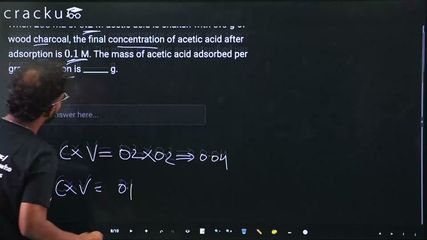
When $$200$$ mL of $$0.2$$ M acetic acid is shaken with $$0.6$$ g of wood charcoal, the final concentration of acetic acid after adsorption is $$0.1$$ M. The mass of acetic acid adsorbed per gram of carbon is ______ g.
Correct Answer: 2
The volume of acetic acid solution is 200 mL = 0.2 L, the initial concentration is 0.2 M, the final concentration after adsorption is 0.1 M, and the mass of wood charcoal is 0.6 g.
Since volume and concentration are known, the initial moles of acetic acid are calculated as:
$$ n_{\text{initial}} = 0.2 \times 0.2 = 0.04 \text{ mol} $$
Similarly, the final moles of acetic acid after adsorption are given by:
$$ n_{\text{final}} = 0.2 \times 0.1 = 0.02 \text{ mol} $$
This gives the moles of acetic acid adsorbed as:
$$ n_{\text{adsorbed}} = 0.04 - 0.02 = 0.02 \text{ mol} $$
Substituting the molar mass of $$CH_3COOH = 60$$ g/mol gives the mass of acetic acid adsorbed:
$$ m_{\text{adsorbed}} = 0.02 \times 60 = 1.2 \text{ g} $$
From the above, the mass of acetic acid adsorbed per gram of carbon is:
$$ \frac{m_{\text{adsorbed}}}{m_{\text{carbon}}} = \frac{1.2}{0.6} = 2 \text{ g/g} $$
Therefore, the mass of acetic acid adsorbed per gram of carbon is 2 g.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
