NTA JEE Mains 8th April 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 8th April 2024 Shift 1 - Question 31
Combustion of glucose $$(C_6H_{12}O_6)$$ produces $$CO_2$$ and water. The amount of oxygen (in g) required for the complete combustion of $$900 \text{ g}$$ of glucose is : [Molar mass of glucose in $$\text{gmol}^{-1} = 180$$]
NTA JEE Mains 8th April 2024 Shift 1 - Question 32
Match List I with List II
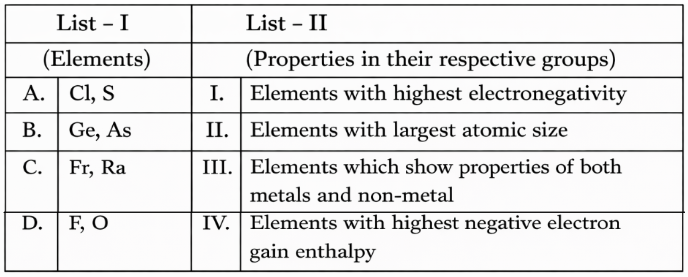
Choose the correct answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Question 33
Match List I with List II
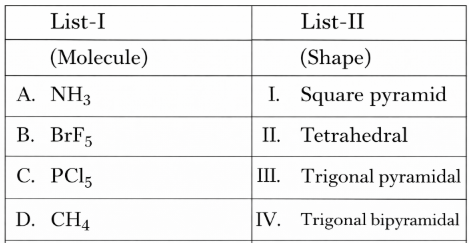
Choose the correct answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Question 34
Match List I with List II
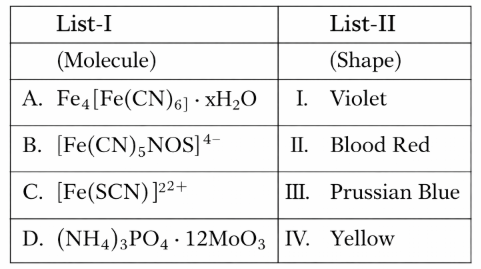
Choose the correct answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Question 35
Given below are two statements: Statement I: $$N(CH_3)_3$$ and $$P(CH_3)_3$$ can act as ligands to form transition metal complexes. Statement II: As N and P are from same group, the nature of bonding of $$N(CH_3)_3$$ and $$P(CH_3)_3$$ is always same with transition metals. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Question 36
For the given hypothetical reactions, the equilibrium constants are as follows: $$X \rightleftharpoons Y; K_1 = 1.0$$, $$Y \rightleftharpoons Z; K_2 = 2.0$$, $$Z \rightleftharpoons W; K_3 = 4.0$$. The equilibrium constant for the reaction $$X \rightleftharpoons W$$ is
NTA JEE Mains 8th April 2024 Shift 1 - Question 37
Among the following halogens $$F_2, Cl_2, Br_2$$ and $$I_2$$. Which can undergo disproportionation reactions?
NTA JEE Mains 8th April 2024 Shift 1 - Question 38
Thiosulphate reacts differently with iodine and bromine in the reactions given below: $$2S_2O_3^{2-} + I_2 \rightarrow S_4O_6^{2-} + 2I^-$$, $$S_2O_3^{2-} + 5Br_2 + 5H_2O \rightarrow 2SO_4^{2-} + 4Br^- + 10H^+$$. Which of the following statement justifies the above dual behaviour of thiosulphate?
NTA JEE Mains 8th April 2024 Shift 1 - Question 39
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R: Assertion A: The stability order of +1 oxidation state of Ga, In and Tl is $$Ga < In < Tl$$. Reason R: The inert pair effect stabilizes the lower oxidation state down the group. In the light of the above statements, choose the correct answer from the options given below:
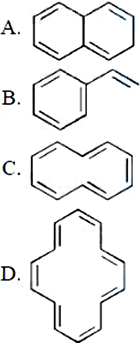
NTA JEE Mains 8th April 2024 Shift 1 - Question 40
Which of the following are aromatic?