NTA JEE Mains 8th April 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 8th April 2024 Shift 1 - Question 41
Given below are two statements:
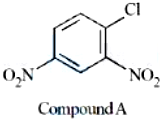
Statement I: IUPAC name of Compound A is 4-chloro-1,3-dinitrobenzene.
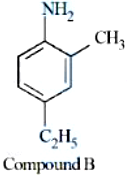
Statement II: IUPAC name of Compound B is 4-ethyl-2-methylaniline. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Question 42
In the given compound, the number of $$2°$$ carbon atom/s is _______.
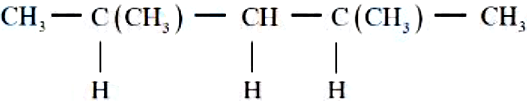
NTA JEE Mains 8th April 2024 Shift 1 - Question 43
Iron (III) catalyses the reaction between iodide and persulphate ions, in which
A. $$Fe^{3+}$$ oxidises the iodide ion
B. $$Fe^{3+}$$ oxidises the persulphate ion
C. $$Fe^{2+}$$ reduces the iodide ion
D. $$Fe^{2+}$$ reduces the persulphate ion.
Choose the most appropriate answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Question 44
Number of Complexes with even number of electrons in $$t_{2g}$$ orbitals is - $$[Fe(H_2O)_6]^{2+}$$, $$[Co(H_2O)_6]^{2+}$$, $$[Co(H_2O)_6]^{3+}$$, $$[Cu(H_2O)_6]^{2+}$$, $$[Cr(H_2O)_6]^{2+}$$
NTA JEE Mains 8th April 2024 Shift 1 - Question 45
An octahedral complex with the formula $$CoCl_3 \cdot nNH_3$$ upon reaction with excess of $$AgNO_3$$ solution gives 2 moles of $$AgCl$$. Consider the oxidation state of $$Co$$ in the complex is '$$x$$'. The value of "$$x + n$$" is ______
NTA JEE Mains 8th April 2024 Shift 1 - Question 46
Which among the following compounds will undergo fastest $$S_N2$$ reaction.
NTA JEE Mains 8th April 2024 Shift 1 - Question 47
Identify the major products A and B respectively in the following set of reactions.
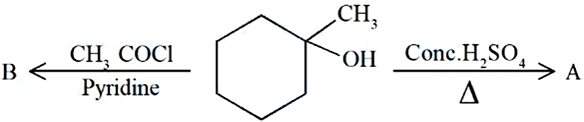
NTA JEE Mains 8th April 2024 Shift 1 - Question 48
Identify the product (P) in the following reaction:
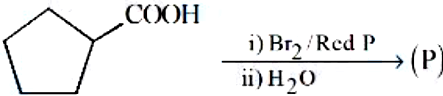
NTA JEE Mains 8th April 2024 Shift 1 - Question 49
Match List I with List II
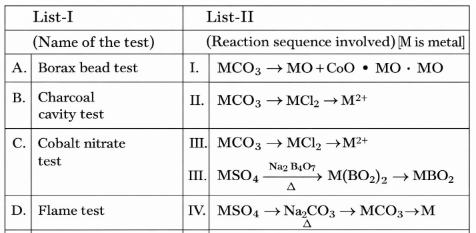
Choose the correct answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Question 50
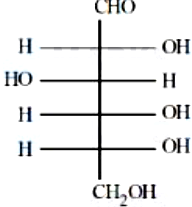
The incorrect statement regarding the given structure is


