NTA JEE Main 27th July 2022 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 31
$$250 \text{ g}$$ solution of D-glucose in water contains $$10.8\%$$ of carbon by weight. The molality of the solution is nearest to (Given: Atomic Weights are $$H = 1u; C = 12u; O = 16u$$)
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 32
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Energy of $$2s$$ orbital of hydrogen atom is greater than that of $$2s$$ orbital of lithium.
Reason R: Energies of the orbitals in the same subshell decrease with increase in the atomic number.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 33
The incorrect statement is
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 34
Given below are two statements.
Statement I: $$O_2$$, $$Cu^{2+}$$ and $$Fe^{3+}$$ are weakly attracted by magnetic field and are magnetized in the same direction as magnetic field.
Statement II: $$NaCl$$ and $$H_2O$$ are weakly magnetized in opposite direction to magnetic field.
In the light of the above statements, choose the most appropriate answer form the options given below
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 35
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Activated charcoal adsorbs $$SO_2$$ more efficiently than $$CH_4$$.
Reason R: Gases with lower critical temperatures are readily adsorbed by activated charcoal.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 36
Given below are two statements:
Statement I: Hydrogen peroxide can act as an oxidizing agent in both acidic and basic conditions.
Statement II: Density of hydrogen peroxide at $$298 \text{ K}$$ is lower than that of $$D_2O$$.
In the light of the above statements. Choose the correct answer from the options
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 37
Given below are two statements
Statement I: The chlorides of Be and Al have Cl-bridged structure. Both are soluble in organic solvents and act as Lewis bases.
Statement II: Hydroxides of Be and Al dissolve in excess alkali to give beryllate and aluminate ions.
In the light of the above statements. Choose the correct answer from the options
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 38
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: [6] Annulene, [8] Annulene and cis-[10] Annulene are respectively aromatic, not-aromatic and aromatic.
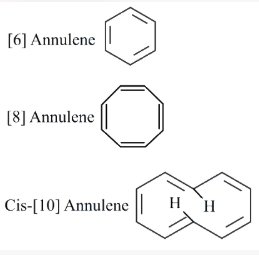
Reason R: Planarity is one of the requirements of aromatic systems.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 39
In Carius method of estimation of halogen, $$0.45 \text{ g}$$ of an organic compound gave $$0.36 \text{ g}$$ of $$AgBr$$. Find out the percentage of bromine in the compound. (Molar masses: $$AgBr = 188 \text{ g mol}^{-1}; Br = 80 \text{ g mol}^{-1}$$)
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 40
Match List I with List II

Choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 41
Boiling point of a $$2\%$$ aqueous solution of a nonvolatile solute A is equal to the boiling point of $$8\%$$ aqueous solution of a non-volatile solute B. The relation between molecular weights of A and B is
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 42
Which of the following methods are not used to refine any metal?
(A) Liquation
(B) Calcination
(C) Electrolysis
(D) Leaching
(E) Distillation
Choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 43
Which oxoacid of phosphorous has the highest number of oxygen atoms present in its chemical formula?
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 44
Given below are two statements:
Statement I: Iron (III) catalyst, acidified $$K_2Cr_2O_7$$ and neutral $$KMnO_4$$ have the ability to oxidise $$I^-$$ to $$I_2$$ independently.
Statement II: Manganate ion is paramagnetic in nature and involves $$p\pi - p\pi$$ bonding.
In the light of the above statements, choose the correct answer from the options.
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 45
The total number of $$Mn = O$$ bonds in $$Mn_2O_7$$ is
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 46
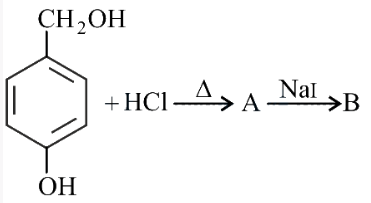
In the above reaction, product B is
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 47
Match List-I with List-II

Choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 48
Match List I with List II

Choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 49
Match List-I with List-II.
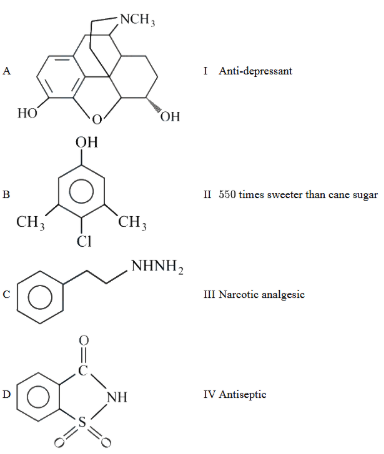
Choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 50
A sugar 'X' dehydrates very slowly under acidic condition to give furfural which on further reaction with resorcinol gives the coloured product after sometime. Sugar 'X' is
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 51
Amongst the following the number of oxide(s) which are paramagnetic in nature is ______
$$Na_2O, KO_2, NO_2, N_2O, ClO_2, NO, SO_2, Cl_2O$$
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 52
According to MO theory, number of species/ions from the following having identical bond order is ______
$$CN^-, NO^+, O_2, O_2^+, O_2^{2+}$$
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 53
The molar heat capacity for an ideal gas at constant pressure is $$20.785 \text{ J K}^{-1} \text{ mol}^{-1}$$. The change in internal energy is $$5000 \text{ J}$$ upon heating it from $$300 \text{ K}$$ to $$500 \text{ K}$$. The number of moles of the gas at constant volume is ______ (Given: $$R = 8.314 \text{ J K}^{-1} \text{ mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 54
At $$310 \text{ K}$$, the solubility of $$CaF_2$$ in water is $$2.34 \times 10^{-3} \text{ g/100 mL}$$. The solubility product of $$CaF_2$$ is ______ $$\times 10^{-8} (\text{mol/L})^3$$ (nearest integer). (Given molar mass: $$CaF_2 = 78 \text{ g mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 55
$$20 \text{ mL}$$ of $$0.02M K_2Cr_2O_7$$ solution is used for the titration of $$10 \text{ mL}$$ of $$Fe^{2+}$$ solution in the acidic medium. The molarity of $$Fe^{2+}$$ solution is ______ $$\times 10^{-2} M$$
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 56
In the following reaction,
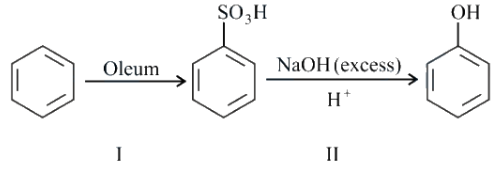
the $$\%$$ yield for reaction I is $$60\%$$ and that of reaction II is $$50\%$$. The overall yield of the complete reaction is ______ $$\%$$.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 57
$$2NO + 2H_2 \rightarrow N_2 + 2H_2O$$. The above reaction has been studied at $$800°C$$.The related data are given in the table below

The order of the reaction with respect to NO is ______.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 58
In the titration of $$KMnO_4$$ and oxalic acid in acidic medium, the change in oxidation number of carbon at the end point is ______.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 59
The conductivity of a solution of complex with formula $$CoCl_3(NH_3)_4$$ corresponds to $$1:1$$ electrolyte, then the primary valency of central metal ion is ______.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Chemistry - Question 60
Optical activity of an enantiomeric mixture is $$+12.6°$$ and the specific rotation of $$(+)$$ isomer is $$+30°$$. The optical purity is ______ $$\%$$.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




