Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The molar heat capacity for an ideal gas at constant pressure is $$20.785 \text{ J K}^{-1} \text{ mol}^{-1}$$. The change in internal energy is $$5000 \text{ J}$$ upon heating it from $$300 \text{ K}$$ to $$500 \text{ K}$$. The number of moles of the gas at constant volume is ______ (Given: $$R = 8.314 \text{ J K}^{-1} \text{ mol}^{-1}$$)
Correct Answer: 2
We need to find the number of moles of gas given the molar heat capacity at constant pressure and the change in internal energy. The following values are provided: $$C_p = 20.785 \text{ J K}^{-1} \text{ mol}^{-1}$$, $$\Delta U = 5000 \text{ J}$$ (change in internal energy), $$T_1 = 300 \text{ K}$$, $$T_2 = 500 \text{ K}$$, and $$R = 8.314 \text{ J K}^{-1} \text{ mol}^{-1}$$.
Since the relation $$C_p - C_v = R$$ holds, substituting the given values gives us $$C_v = C_p - R = 20.785 - 8.314 = 12.471 \text{ J K}^{-1} \text{ mol}^{-1}$$.
For an ideal gas at constant volume, the change in internal energy is given by $$\Delta U = n C_v \Delta T$$.
From the temperatures provided, $$\Delta T = T_2 - T_1 = 500 - 300 = 200 \text{ K}$$. Substituting both $$\Delta T$$ and the value of $$C_v$$ into the energy equation gives $$5000 = n \times 12.471 \times 200$$, which simplifies to $$5000 = n \times 2494.2$$. Solving for $$n$$ yields $$n = \frac{5000}{2494.2} = 2.005 \approx 2$$.
The correct answer is 2.
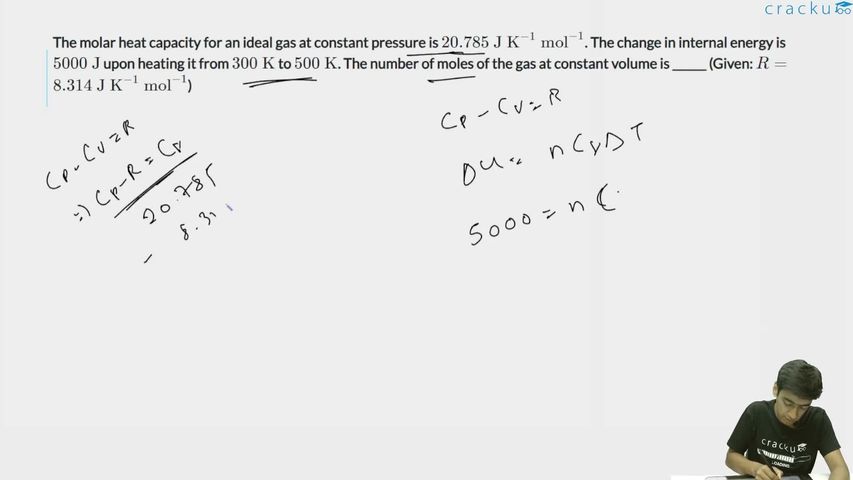
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
