Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
According to MO theory, number of species/ions from the following having identical bond order is ______
$$CN^-, NO^+, O_2, O_2^+, O_2^{2+}$$
Correct Answer: 3
We need to find how many species/ions from the given list have identical bond order using Molecular Orbital theory.
Bond order = $$\frac{N_b - N_a}{2}$$ where $$N_b$$ is the number of bonding electrons and $$N_a$$ is the number of antibonding electrons.
For $$CN^-$$, the total number of electrons is 6 + 7 + 1 = 14, and its molecular orbital configuration is $$(\sigma_{1s})^2(\sigma^*_{1s})^2(\sigma_{2s})^2(\sigma^*_{2s})^2(\pi_{2p})^4(\sigma_{2p})^2$$. Substituting into the bond order formula gives $$\frac{10 - 4}{2} = 3$$.
Since $$NO^+$$ is isoelectronic with $$CN^-$$ (7 + 8 − 1 = 14 electrons), it has the same MO configuration and therefore the same bond order: $$\frac{10 - 4}{2} = 3$$.
For $$O_2$$, which has 16 electrons (8 + 8), the MO configuration is $$(\sigma_{1s})^2(\sigma^*_{1s})^2(\sigma_{2s})^2(\sigma^*_{2s})^2(\sigma_{2p})^2(\pi_{2p})^4(\pi^*_{2p})^2$$, yielding a bond order of $$\frac{10 - 6}{2} = 2$$.
When one electron is removed to form $$O_2^+$$ (16 − 1 = 15 electrons), the configuration becomes $$(\sigma_{1s})^2(\sigma^*_{1s})^2(\sigma_{2s})^2(\sigma^*_{2s})^2(\sigma_{2p})^2(\pi_{2p})^4(\pi^*_{2p})^1$$, giving a bond order of $$\frac{10 - 5}{2} = 2.5$$.
Removing two electrons to obtain $$O_2^{2+}$$ (16 − 2 = 14 electrons) leads to the configuration $$(\sigma_{1s})^2(\sigma^*_{1s})^2(\sigma_{2s})^2(\sigma^*_{2s})^2(\sigma_{2p})^2(\pi_{2p})^4$$, and the bond order is $$\frac{10 - 4}{2} = 3$$.
From the above calculations, $$CN^-$$, $$NO^+$$, and $$O_2^{2+}$$ each have a bond order of 3. Therefore, the number of species with identical bond order is 3.
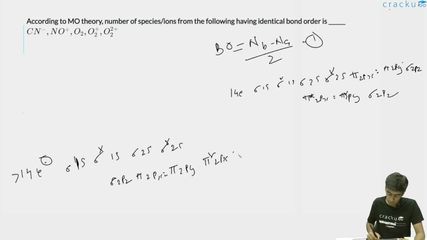
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
