Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
In the following reaction,
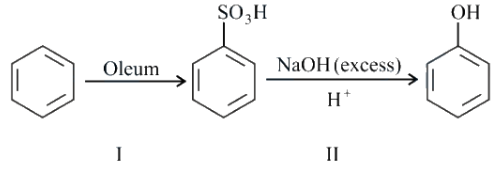
the $$\%$$ yield for reaction I is $$60\%$$ and that of reaction II is $$50\%$$. The overall yield of the complete reaction is ______ $$\%$$.
Correct Answer: 30
We are given a two-step reaction where the yield of reaction I is 60% and the yield of reaction II is 50%.
The overall yield of a multi-step reaction is the product of the individual yields of each step.
Overall yield = Yield of Step I $$\times$$ Yield of Step II
$$\text{Overall yield} = \frac{60}{100} \times \frac{50}{100} = 0.6 \times 0.5 = 0.3$$
Converting to percentage:
$$\text{Overall yield} = 0.3 \times 100 = 30\%$$
Therefore, the overall yield of the complete reaction is $$\textbf{30}$$ %.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
